ICU Management & Practice, Volume 18 - Issue 2, 2018
Detecting pre-hospital trauma-induced coagulopathy
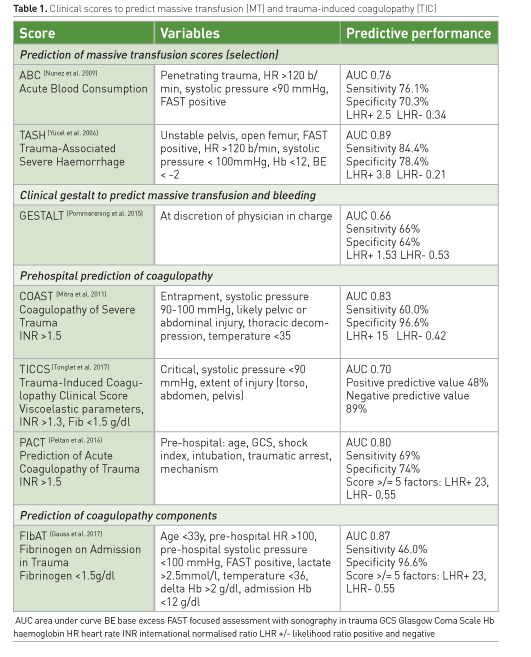
Managing pre-hospital trauma-induced coagulopathy
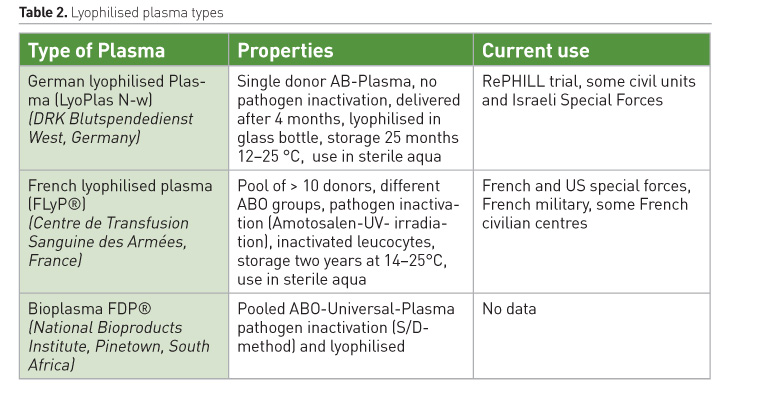
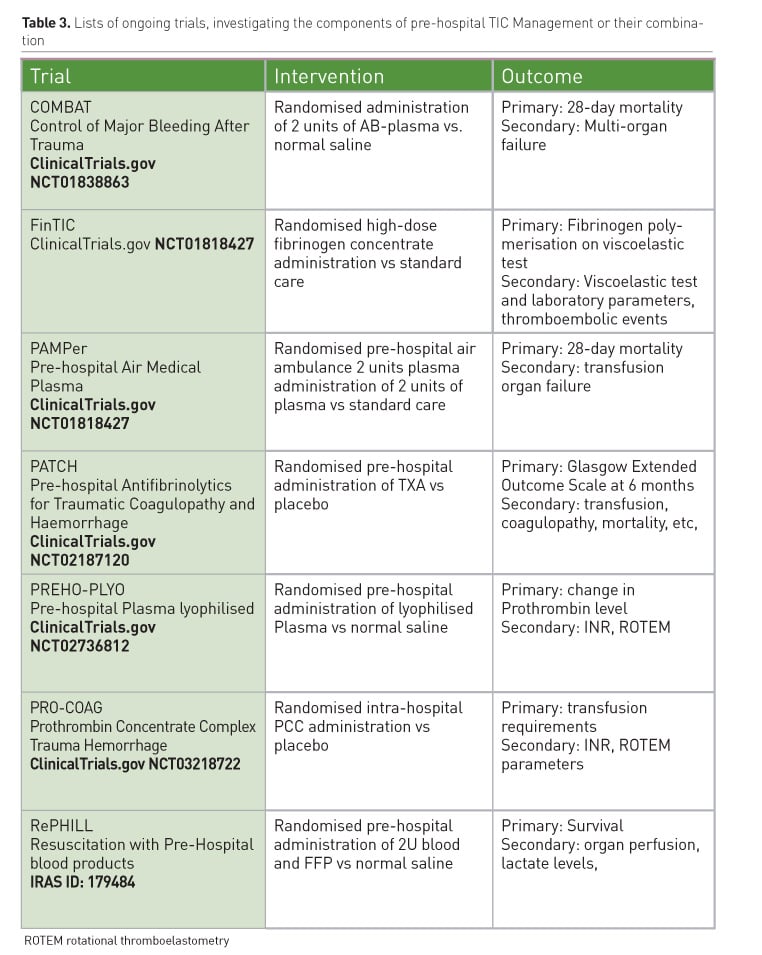
Pre-hospital TIC strategy
Conflict of interest
Abbreviations
ATC acute trauma coagulopathy
FC fibrinogen concentrate
FFP fresh frozen plasma
INR international normalised ratio
PCC prothrombin complex concentrate
POC point-of-care
RBC red blood cells
RC resuscitation coagulopathy
TIC trauma-induced coagulopathy
TXA tranexamic acid
References:
Brockamp T, Nienaber U, Mutschler M et al.; TraumaRegister DGU (2012) Predicting on-going hemorrhage and transfusion requirement after severe trauma: a validation of six scoring systems and algorithms on the TraumaRegister DGU. Crit Care, 16: R129.
Brown JB, Cohen MJ, Minei JP et al. (2015) Pretrauma center red blood cell transfusion is associated with reduced mortality and coagulopathy in severely injured patients with blunt trauma. Ann Surg, 261: 997–1005.
Cantle PM, Cotton BA (2017) Prediction of massive transfusion in trauma. Crit Care Clin, 33: 71–84.
CRASH-2 trial collaborators; Shakur H, Roberts I, Bautista R et al. (2010) Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2): a randomised, placebo-controlled trial. Lancet, 376: 23–32.
Floccard B, Rugeri L, Faure A et al. (2012) Early coagulopathy in trauma patients: an on-scene and hospital admission study. Injury, 43: 26–32.
Garrigue D, Godier A, Glacet A et al. (2018) French lyophilized plasma versus fresh frozen plasma for the initial management of trauma-induced coagulopathy: a randomized open-label trial. J Thromb Haemost, 16: 481–9.
Gauss T, Hamada S, Jurcisin I et al. (2014) Limits of agreement between measures obtained from standard laboratory and the point-of-care device Hemochron Signature Elite ® during acute haemorrhage. Br J Anaesth, 112: 514–20.
Gauss T, Campion S, Kerever S et al.; Traumabase© Group (2017) Fibrinogen on Admission in Trauma score: early prediction of low plasma fibrinogen concentrations in trauma patients. Eur J Anaesthesiol, 35(1):25-32.
Gayet-Ageron A, Prieto-Merino D, Ker K et al.; Antifibrinolytic Trials Collaboration (2018). Effect of treatment delay on the effectiveness and safety of antifibrinolytics in acute severe haemorrhage: a meta-analysis of individual patient-level data from 40 138 bleeding patients. Lancet, 391: 125–32.
Gonzalez E, Moore EE, Moore HB et al. (2016) Goal-directed hemostatic resuscitation of trauma-induced coagulopathy: a pragmatic randomized clinical trial comparing a viscoelastic assay to conventional coagulation assays. Ann Surg, 263: 1051–9.
Grottke O, Levy JH (2015) Prothrombin complex concentrates in trauma and perioperative bleeding. Anesthesiology, 122: 923–31.
Hagemo JS (2013) Prehospital detection of traumatic coagulopathy. Transfusion, 53 Suppl 1: 48S-51S.
Hamada SR, Gauss T, Pann J et al. (2015) European trauma guideline compliance assessment: the ETRAUSS study. Crit Care, 19: 423.
Hansson KM, Johansson KJ, Wingren C et al. (2017) Recombinant human prothrombin reduced blood loss in a porcine model of dilutional coagulopathy with uncontrolled bleeding. Blood Coagul Fibrinolysis, 28: 244–53.
Harris T, Davenport R, Mak M et al. (2018) The evolving science of trauma resuscitation. Emerg Med Clin North Am, 36: 85–106.
Hay SI, Abajobir AA, Abate KH et al. (2017) Global, regional, and national disability-adjusted life-years (DALYs) for 333 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet, 390: 1260–344.
Holcomb JB, Tilley BC, Baraniuk S et al. (2015) Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma: the PROPPR randomized clinical trial. JAMA, 313: 471–82.
Inaba K, Karamanos E, Lustenberger T et al. (2013) Impact of fibrinogen levels on outcomes after acute injury in patients requiring a massive transfusion. J Am Coll Surg, 216: 290–7.
Innerhofer P, Westermann I, Tauber H et al. (2013) The exclusive use of coagulation factor concentrates enables reversal of coagulopathy and decreases transfusion rates in patients with major blunt trauma. Injury, 44: 209–16.
Jenkins D, Stubbs J, Williams S et al. (2014) Implementation and execution of civilian remote damage control resuscitation programs. Shock, 41 Suppl 1: 84–9.
Johansson PI, Stensballe J, Ostrowski SR (2017) Shock induced endotheliopathy (SHINE) in acute critical illness - a unifying pathophysiologic mechanism. Critical Care, 21(1): 25.
Khan S, Brohi K, Chana M et al.; International Trauma Research Network (INTRN) (2014) Hemostatic resuscitation is neither hemostatic nor resuscitative in trauma hemorrhage. J Trauma Acute Care Surg, 76: 561–7; discussion 567-568.
Kozar RA, Peng Z, Zhang R et al. (2011) Plasma restoration of endothelial glycocalyx in a rodent model of hemorrhagic shock. Anesth Analg, 112: 1289–95.
Kushimoto S, Kudo D, Kawazoe Y (2017) Acute traumatic coagulopathy and trauma-induced coagulopathy: an overview. J Intensive Care, 5: 6.
Lockey DJ, Weaver AE, Davies GE (2013) Practical translation of hemorrhage control techniques to the civilian trauma scene. Transfusion, 53 Suppl 1: 17S-22S.
Lyon RM, de Sausmarez E, McWhirter E et al.; Kent, Surrey & Sussex Air Ambulance Trust (2017) Pre-hospital transfusion of packed red blood cells in 147 patients from a UK helicopter emergency medical service. Scand J Trauma Resusc Emerg Med, 25: 12.
Maegele M, Zinser M, Schlimp C et al. (2015) Injectable hemostatic adjuncts in trauma: Fibrinogen and the FIinTIC study. Journal of Trauma and Acute Care Surgery, 78: S76–S82.
Martinaud C, Civadier C, Ausset S et al. (2012) In vitro hemostatic properties of French lyophilized plasma. Anesthesiology, 117: 339–46.
McQuilten ZK, Wood EM, Bailey M et al. (2017) Fibrinogen is an independent predictor of mortality in major trauma patients: a five-year statewide cohort study. Injury, 48: 1074–81.
Mistral T, Boué Y, Bosson JL et al. (2017) Performance of point-of-care international normalized ratio measurement to diagnose trauma-induced coagulopathy. Scand J Trauma Resusc Emerg Med, 25(1): 59.
Mitra B, Cameron PA, Mori A et al. (2011) Early prediction of acute traumatic coagulopathy. Resuscitation, 82: 1208–13.
Moore EE, Moore HB, Gonzalez E et al. (2016) Rationale for the selective administration of tranexamic acid to inhibit fibrinolysis in the severely injured patient. Transfusion, 56 Suppl 2: S110-114.
Morrison JJ, Dubose JJ, Rasmussen TE et al. (2012) Military application of tranexamic acid in trauma emergency resuscitation (MATTERs) Study. Arch Surg, 147: 113–9.
Nunez TC, Voskresensky IV, Dossett LA et al. (2009). Early prediction of massive transfusion in trauma: simple as ABC (assessment of blood consumption)? J Trauma, 66: 346–52.
Ostrowski SR, Sørensen AM, Larsen CF et al. (2011) Thrombelastography and biomarker profiles in acute coagulopathy of trauma: a prospective study. Scand J Trauma Resusc Emerg Med, 19: 64.
Ostrowski SR, Henriksen HH, Stensballe J et al. (2017) Sympathoadrenal activation and endotheliopathy are drivers of hypocoagulability and hyperfibrinolysis in trauma: a prospective observational study of 404 severely injured patients. J Trauma Acute Care Surg, 82: 293–301.
Oyeniyi BT, Fox EE, Scerbo M et al. (2017) Trends in 1029 trauma deaths at a level 1 trauma center: Impact of a bleeding control bundle of care. Injury, 48: 5–12.
Peltan ID, Rowhani-Rahbar A, Vande Vusse LK et al. (2016) Development and validation of a prehospital prediction model for acute traumatic coagulopathy. Crit Care, 20: 371.
Pommerening MJ, Goodman MD, Holcomb JB et al. (2015) Clinical gestalt and the prediction of massive transfusion after trauma. Injury 46: 807–13.
Rehn M, Weaver A, Brohi K et al. (2018) Effect of pre-hospital red blood cell transfusion on mortality and time of death in civilian trauma patients. Shock, Apr 18.
Schöchl H, Nienaber U, Hofer G et al. (2010) Goal-directed coagulation management of major trauma patients using thromboelastometry (ROTEM)-guided administration of fibrinogen concentrate and prothrombin complex concentrate. Crit Care, 14: R55.
Schöchl H, Schlimp CJ, Maegele M (2014) Tranexamic acid, fibrinogen concentrate, and prothrombin complex concentrate: data to support prehospital use? Shock, 41 Suppl 1: 44–6.
Smith IM, James RH, Dretzke J et al. (2016) Prehospital blood product resuscitation for trauma: a systematic review. Shock, 46: 3–16.
Spinella PC, Cap AP (2017) Prehospital hemostatic resuscitation to achieve zero preventable deaths after traumatic injury. Curr Opin Hematol, 24: 529–35.
Spinella PC, Pidcoke HF, Strandenes G et al. (2016) Whole blood for hemostatic resuscitation of major bleeding. Transfusion, 56 Suppl 2: S190-202.
Stanworth SJ, Davenport R, Curry N et al. (2016) Mortality from trauma haemorrhage and opportunities for improvement in transfusion practice. Br J Surg, 103: 357–65.
Stinger HK, Spinella PC, Perkins JG et al. (2008) The ratio of fibrinogen to red cells transfused affects survival in casualties receiving massive transfusions at an army combat support hospital. J Trauma 64: S79-85; discussion S85.
Tien HC, Spencer F, Tremblay LN et al. (2007) Preventable deaths from hemorrhage at a level I Canadian trauma center. J Trauma, 62, 142–6.
Tonglet M, Lefering R, Minon JM et al. (2017) Prehospital identification of trauma patients requiring transfusion: results of a retrospective study evaluating the use of the trauma induced coagulopathy clinical score (TICCS) in 33,385 patients from the TraumaRegister DGU®. Acta Chir Belg, 117: 385–90.
Wafaisade A, Lefering R, Bouillon Bet al. (2016) Prehospital administration of tranexamic acid in trauma patients. Crit Care, 20: 143.
Weaver AE, Thompson J, Lockey DJ (2012) The effectiveness of a simple ‘Code Red’ transfusion request policy initiated by pre-hospital physicians. Scand J Trauma Resusc Emerg Med., 20(Suppl 1): O1.
Yücel N, Lefering R, Maegele M et al. (2006) Trauma associated severe hemorrhage (TASH)-score: probability of mass transfusion as surrogate for life threatening hemorrhage after multiple trauma. J Trauma, 60: 1228–36; discussion 1236-7.






















