ICU Management & Practice, Volume 16 - Issue 4, 2016
Severe hypoxaemia and cardiovascular collapse, leading to cardiac arrest, cerebral anoxia and death, are the most frequent complications related to intubation in intensive care units (ICU), associated with difficulty of intubation. To prevent and limit the incidence of difficult intubation, specific risk factors for difficult intubation in the ICU have been identified and pre-oxygenation techniques and intubation algorithms have been developed. The objectives of this review are to describe new tools (i.e. MACOCHA Score) to better identify patients at high risk for difficult intubation and new strategies for improving preoxygenation before intubation and decreasing difficult intubation incidence, using specific algorithms.
Airway management is one of the most commonly performed procedures in the intensive care unit (ICU). Difficult intubation incidence is 10% on average (De Jong et al. 2013b), ranging from 1% to 23% depending on the centre and the definition used (Martin et al. 2011; Heuer et al. 2012; Simpson et al.; 2012; Le Tacon et al. 2000). Hypoxaemia and cardiovascular collapse represent the initial and most serious life-threatening complications associated with difficult airway access in the critically ill (Jaber et al. 2006; De Jong et al. 2014a; 2015). To prevent and limit the incidence of life-threatening complications following difficult intubation, a score that allows accurate prediction of difficult intubation in the ICU has been developed. In addition, several preoxygenation techniques and intubation algorithms are outlined, including new devices for intubation such as videolaryngoscopes.
Patients At Risk of Difficult Intubation
We assessed for the first time in a large prospective cohort the risk factors of difficult intubation in the ICU (De Jong et al. 2013b). A predictive score of difficult intubation, the MACOCHA score, was developed and then externally validated.
The main predictors of difficult intubation were related to the patient (Mallampati score III or IV, obstructive apnoea syndrome, reduced mobility of cervical spine, limited mouth opening), the pathology (coma, severe hypoxia) and the operator (non-anaesthesiologist) (Table 1) (De Jong et al. 2013b; De Jong et al. 2014a). In order to reject difficult intubation with certainty, a cutoff of 3 or greater was appropriate, allowing optimal negative predictive value (respectively 97% and 98% in the original and validation cohorts) and sensitivity (respectively 76% and 73% in the original and validation cohorts). The MACOCHA score allows identification of patients at risk of difficult intubation.

All ICU patients could be considered at risk of complications of intubation, and particularly those with difficult intubation. Anticipating difficult intubation is a challenging issue: the complications of intubation are higher in cases of difficult intubation when compared to non difficult intubation (65% vs. 41% overall for both moderate and severe complications, 51% vs. 36% for severe life-threatening complications) (De Jong et al. 2013b).
Intubation Recommendations Bundle to Limit Complications Related to Intubation Procedure (The Montpellier-ICU Intubation Algorithm)
In order to reduce the complications associated with all intubation in the ICU and in particular difficult intubation, a bundle of techniques has been developed for ICUs and is presented in Table 2 (Jaber et al. 2010; De Jong et al. 2014a; Jaber et al. 2016).

Preoxygenation and recruitment manoeuvres (RM) are one of the procedures that may improve airway safety. Figure 1 summarises the minimal SpO2 values during the intubation procedure in severe hypoxaemic patients throughout both randomised and non-randomised studies according to the method of preoxygenation (Jaber et al. 2016).
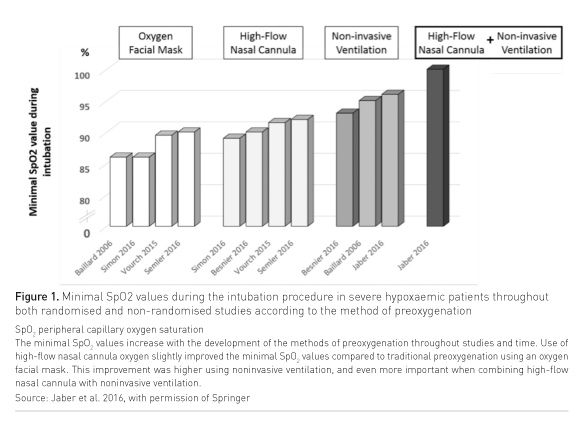
Several manoeuvres in spontaneous ventilation (e.g. 3-8 vital capacities vs 3 minutes tidal volume breathing) exist and seem to be almost equally effective (Tanoubi et al 2009). Some technical details, however, can make a significant difference. First, the clinician needs to make sure the facemask properly fits the patient’s facial morphology. Second, fresh gas flow needs to be set at a high range to homogenise ventilation through the lungs and to decrease the impact of leaks. Third, leaks should be avoided and diagnosed either by a flaccid reservoir bag or by the absence of a normal capnography waveform, since leaks impair the efficacy of preoxygenation. In critically ill patients, the advantage of a prolonged period of preoxygenation has not been clearly demonstrated. Most of them present with acute respiratory failure with a certain amount of shunt, a reduced functional reserve capacity, and do not respond to administration of oxygen as well as patients scheduled for surgery, even after an increase of preoxygenation duration from 4 to 8 minutes (Mort et al. 2009).
Noninvasive ventilation (NIV) as a preoxygenation manoeuvre has therefore been evaluated in critically ill patients. The rationale for use of NIV during preoxygenation is to recruit lung tissue available for gas exchange: to “open the lung” with the pressure support and “keep the lung open” with the positive end-expiratory pressure (PEEP), which permits limitation of alveolar derecruitment. Our group reported its benefits compared to administration of oxygen alone (Baillard et al. 2006). Indeed, in a randomized controlled trial including hypoxaemia patients, the incidence of severe hypoxaemia (peripheral saturation in oxygen [SpO2] below 80%) within 30 minutes after intubation was 7% in the NIV group, compared to 42% in the oxygen group. To perform NIV for 3 to 5 minutes in critically ill patients, the facial masks available in every ICU room are sufficient. The patient should be in the semi-sitting position, FiO2 set at 100%, inspiratory pressure set to observe a tidal volume of 6 to 10 ml/kg and respiratory rate of 10 to 25 cycles/minute. However, NIV has to be removed during the tracheal intubation, and no oxygen is administered to the patient during this apnoea period (Mosier et al. 2016; Papazian et al. 2016).
Recently we reported the first trial assessing preoxygenation using a novel strategy of preoxygenation adding apnoeic high-flow nasal cannula oxygen (HFNC) to NIV to perform orotracheal intubation in hypoxaemic ICU patients (Jaber et al. 2016). We found that, despite similar baseline characteristics and oxygenation, this novel strategy of preoxygenation was more effective at reducing oxygen desaturation in comparison to the reference method using NIV alone. This proof-of-concept study suggests that the novel strategy of preoxygenation using the association of NIV and HFNC for apnoeic oxygenation applied together is safe and more effective in preventing oxygen desaturation than the preoxygenation reference method using NIV alone in critically ill patients with severe hypoxaemic respiratory failure. This novel approach could be used in severe hypoxaemic ICU patients in order to decrease severe hypoxaemia related to intubation procedure, which can result in cardiac arrest, cerebral anoxia and death.
To open the lung after the apnoea period during intubation procedure, an RM, which consists of a transient increase in inspiratory pressure, can be applied after tracheal intubation. Several manoeuvres exist, but the one best described in this situation consists of applying a continuous positive airway pressure (CPAP) of 40cmH2O for 30 to 40 seconds (Constantin et al. 2010; Futier et al. 2011; 2010). A randomized controlled trial was conducted by our group in 40 critically ill patients in the ICU who required intubation for acute hypoxaemic respiratory failure (Constantin et al. 2010). Compared to no RM, RM performed immediately after intubation was associated with a higher PaO2 (under 100% FiO2) both 5 minutes after intubation (93 ± 36 vs 236 ± 117 mmHg) and 30 minutes after intubation (110 ± 39 and 180 ± 79 mmHg).
Preoxygenation and RM are only one of the procedures that may improve airway safety. Managing the airway of at-risk patients presents some unique challenges for the anaesthesiologist or intensivist. The combination of a limited physiologic reserve in these patients and the potential for difficult mask ventilation and intubation mandates careful planning with a good working knowledge of alternative tools and strategies, should conventional attempts at securing the airway fail. To limit the incidence of severe complications occurring after this potentially hazardous procedure, the whole process (pre-, per- and post-intubation) should be guided by protocols geared towards patients’ safety. In the ICU we designed a multicentre study and described how implementation of such bundle protocols improves the safety of airway management (Jaber et al. 2010). This bundle, the Montpellier ICU intubation algorithm, is summarised in Table 2 (De Jong et al. 2014a; Jaber et al. 2010; 2016).
Briefly, pre-intubation period interventions consist of fluid loading in the absence of cardiogenico oedema, preoxygenation with NIV and HFNC for apnoeic oxygenation in the case of acute respiratory failure, preparation of sedation by the nursing team and the presence of two operators.
During the intubation period, recommended induction is rapid sequence induction (RSI) using short acting, well-tolerated hypnotics (etomidate or ketamine), and a rapid onset muscle relaxant (succinylcholine), with application of cricoids pressure (Sellick manoeuvre).
Just after the intubation (post-intubation period), we recommend verification of the tube’s position by capnography (a technique which allows confirmation of the endotracheal position of the tube and verification of the absence of oesophageal placement), initiation of long-term sedation as soon as possible (to avoid agitation) and use of protective mechanical ventilation settings, with a RM following intubation after haemodynamic stabilisation. At any time, vasopressors are mandatory in the event of severe haemodynamic collapse.
Airway Management
Algorithm for Difficult Intubation in the ICU
As previously recommended in the operating theatre (Amathieu et al. 2011), an airway management algorithm is advised in the ICU, as proposed in Figure 2 (De Jong et al. 2014a).
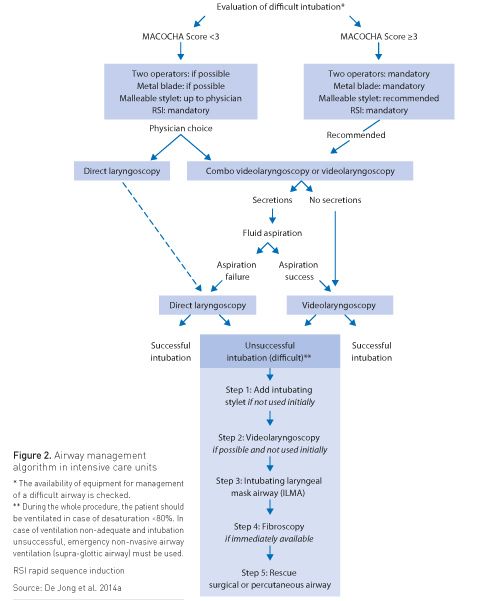
First, the difficulty of intubation is evaluated using the MACOCHA score (De Jong et al. 2013b). The availability of equipment for management of a difficult airway is checked.
During the procedure, the patient should be ventilated in case of desaturation <80%. In case of inadequate ventilation and unsuccessful intubation, emergency noninvasive airway ventilation (supra-glottic airway) must be used.
Then, if difficult intubation is predicted (MACOCHA score ≥3), the presence of two operators and a metal blade is advised, and use of a malleable stylet is recommended. Videolaryngoscopy or combo videolaryngoscopy are also recommended in case of predicted difficult intubation. Otherwise, the choice of the device is at the discretion of the physician. In case of abundant secretions even after aspiration, direct laryngoscopy is preferred rather than videolaryngoscopy. Videolaryngosopes are indirect rigid fibre optic laryngoscopes with a video camera mounted at the end of an angled blade. Recently, videolaryngoscopes, such as C-Mac® (Karl Storz) (Noppens et al. 2012; Ng et al. 2012) or GlideScope (Verathon) (Kory et al. 2013; Lakticova et al. 2013) have demonstrated their efficiency in the ICU setting. Moreover, a recent study (De Jong et al. 2013a) assessed a new mixed videolaryngoscope that can be used as a direct or indirect view laryngoscope. This before-after prospective study showed that the systematic use of a mixed videolaryngoscope for intubation in a quality improvement process using an airway management algorithm significantly reduces the incidence of difficult laryngoscopy and/or difficult intubation.
These results were confirmed by a systematic review and meta-analysis establishing that use of videolaryngoscopes for intubation in the ICU could reduce the rate of difficult intubation (De Jong et al. 2014b). When used by trained operators, videolaryngoscopes seem efficient to reduce difficult intubation in ICU, but a large multicentre study is needed to assess if the complications of intubation are decreased using videolaryngoscopes.
Finally, in case of intubation failure, an intubating stylet (malleable stylet or long flexible angulated stylet) is added first, followed successively by the use of videolaryngoscopy if not used first, the use of an intubation laryngeal mask airway, the use of fibroscopy and finally the use of a rescue percutaneous or surgical airway.
Studies are needed to assess if applying this protocol in Ithe ICU leads to a reduction of difficult intubations and complications. In each ICU, this airway management algorithm should be adapted according to the ICU ways of working.
Conclusion
Predicting risk factors of difficult intubation should be assessed in the ICU, in order to identify the patients at risk of difficult intubation using a simple score applicable at the bedside, the MACOCHA score. Preoxygenation is a standard of care before intubation, with the aim of increasing the lung stores of oxygen. In critically ill patients, the combination of NIV, HFNC to allow apnoeic oxygenation and post-intubation RM is recommended. Then an intubation bundle should be applied in order to reduce complications of difficult intubation, including verification of the endotracheal position of the tube using capnography. Finally, an airway management algorithm is strongly advised in the ICU, adapted according to the ICU’s ways of working and the training of operators for new intubation devices such as videolaryngoscopes.
References:
Amathieu, R, Combes X, Abdi W et al. (2011. An algorithm for difficult airway management, modified for modern optical devices (Airtraq laryngoscope; LMA CTrach™): a 2-year prospective validation in patients for elective abdominal, gynecologic, and thyroid surgery. Anesthesiology, 114(1): 25-33.
PubMed ↗
Baillard C, Fosse JP, Sebbane M et al. (2006) Noninvasive ventilation improves preoxygenation before intubation of hypoxic patients. Am J Respir Crit Care Med, 174(2): 171-7.
PubMed ↗
Constantin JM, Futier E, Cherprenet AL et al. (2010) A recruitment maneuver increases oxygenation after intubation of hypoxemic intensive care unit patients: a randomized controlled study. Crit Care, 14(2): R76.
PubMed ↗
De Jong A, Clavieras N, Conseil M et al. (2013a) Implementation of a combo videolaryngoscope for intubation in critically ill patients: a before-after comparative study. Intensive Care Med, 39(12): 2144-52.
PubMed ↗
De Jong A, Jung B, Jaber S (2014a) Intubation in the ICU: we could improve our practice. Crit Care, 18(2): 209.
PubMed ↗
De Jong A, Molinari N, Conseil M et al. (2014b) Video laryngoscopy versus direct laryngoscopy for orotracheal intubation in the intensive care unit: a systematic review and meta-analysis. Intensive Care Med, 40(5): 629-39.
PubMed ↗
De Jong A, Molinari N, Pouzeratte Y et al. (2015) Difficult intubation in obese patients: incidence, risk factors, and complications in the operating theatre and in intensive care units. Br J Anaesth, 114(2): 297-306.
PubMed ↗
De Jong A, Molinari N, Terzi N et al. (2013b) Early identification of patients at risk for difficult intubation in the intensive care unit: development and validation of the MACOCHA score in a multicenter cohort study. Am J Respir Crit Care Med, 187(8): 832-9.
PubMed ↗
Futier E, Constantin JM, Pelosi P et al. (2011) Noninvasive ventilation and alveolar recruitment maneuver improve respiratory function during and after intubation of morbidly obese patients: a randomized controlled study. Anesthesiology, 114(6): 1354-63.
PubMed ↗
Futier E, Constantin JM, Petit A et al. (2010) Positive end-expiratory pressure improves end-expiratory lung volume but not oxygenation after induction of anaesthesia. Eur J Anaesthesiol, 27(6): 508-13.
PubMed ↗
Heuer JF, Barwing TA, Barwing J et al. (2012) Incidence of difficult intubation in intensive care patients: analysis of contributing factors. Anaesth Intensive Care, 40(1): 120-7.
PubMed ↗
Jaber S, Amraoui J, Lefrant JY et al. (2006) Clinical practice and risk factors for immediate complications of endotracheal intubation in the intensive care unit: a prospective, multiple-center study. Crit Care Med, 34(9): 2355-61.
PubMed ↗
Jaber S, Jung B, Corne P et al. (2010) An intervention to decrease complications related to endotracheal intubation in the intensive care unit: a prospective, multiple-center study. Intensive Care Medicine, 36(2): 248-55.
PubMed ↗
Jaber S, Monnin M, Girard M et al. (2016) Apnoeic oxygenation via high-flow nasal cannula oxygen combined with non-invasive ventilation preoxygenation for intubation in hypoxaemic patients in the intensive care unit: the single-centre, blinded, randomised controlled OPTINIV trial. Intensive Care Med, Oct 11 [Epub ahead of print]
PubMed ↗
Kory P, Guevarra K, Mathew JP et al. (2013) The impact of video laryngoscopy use during urgent endotracheal intubation in the critically ill. Anesth Analg, 117(1); 144-9.
PubMed ↗
Lakticova V, Koenig SJ, Narasimhan M. et al. (2015) Video laryngoscopy is associated with increased first pass success and decreased rate of esophageal intubations during urgent endotracheal intubation in a medical intensive care unit when compared to direct laryngoscopy. J Intensive Care Med, 30(1):44-8.
PubMed ↗
Le Tacon S, Wolter P, Rusterholtz T et al. (2000) [Complications of difficult tracheal intubations in a critical care unit] [Article in French] Ann Fr Anesth Reanim, 19(10): 719-24.
PubMed ↗
Martin LD, Mhyre JM, Shanks AM et al. (2011) 3,423 Emergency tracheal intubations at a university hospital: airway outcomes and complications. Anesthesiology, 114(1): 42-8.
PubMed ↗
Mort TC, Waberski BH, Clive J (2009) Extending the preoxygenation period from 4 to 8 mins in critically ill patients undergoing emergency intubation. Crit Care Med, 37(1): 68-71.
PubMed ↗
Mosier JM, Hypes CD, Sakles JC (2016) Understanding preoxygenation and apneic oxygenation during intubation in the critically ill. Intensive Care Med, Jun 24. [Epub ahead of print]
PubMed ↗
Ng I, Hill AL, Williams DL et al. (2012) Randomized controlled trial comparing the mcgrath videolaryngoscope with the c-mac videolaryngoscope in intubating adult patients with potential difficult airways. Br J Anaesth, 109(3): 439-43.
PubMed ↗
Noppens RR, Geimer S, Eisel N et al. (2012) Endotracheal intubation using the C-MAC® video laryngoscope or the Macintosh laryngoscope: a prospective, comparative study in the ICU. Crit Care, 16(3): R103.
PubMed ↗
Papazian L, Corley A, Hess D et al. (2016) Use of high-flow nasal cannula oxygenation in ICU adults: a narrative review. Intensive Care Med, 42(9): 1336-49.
PubMed ↗
Simpson GD, Ross MJ, McKeown DW et al. (2012) Tracheal intubation in the critically ill: a multi-centre national study of practice and complications. Br J Anaesth, 108(5): 792-9.
PubMed ↗
Tanoubi I, Drolet P, Donati F (2009) Optimizing preoxygenation in adults. Can J Anaesth, 56(6): 449-66.
PubMed ↗






















