3D printing can now be used to print lithium-ion microbatteries the size of a grain of sand. The printed microbatteries could supply electricity to tiny devices in fields from medicine to communications, including many that have lingered on lab benches for lack of a battery small enough to fit the device, yet provide enough stored energy to power them.
To make the microbatteries, a team based at Harvard University and
the University of Illinois at Urbana-Champaign printed precisely
interlaced stacks of tiny battery electrodes, each less than the width
of a human hair.
"Not only did we demonstrate for the first time that we can 3D-print a
battery, we demonstrated it in the most rigorous way,"said Jennifer
Lewis, Ph.D., senior author of the study, who is also the Hansjörg Wyss
Professor of Biologically Inspired Engineering at the Harvard School of
Engineering and Applied Sciences (SEAS), and a Core Faculty Member of
the Wyss Institute for Biologically Inspired Engineering at Harvard
University. Lewis led the project in her prior position at the
University of Illinois at Urbana-Champaign, in collaboration with
co-author Shen Dillon, an Assistant Professor of Materials Science and
Engineering there.
The results were published in the June 18, 2013 online edition of Advanced Materials.
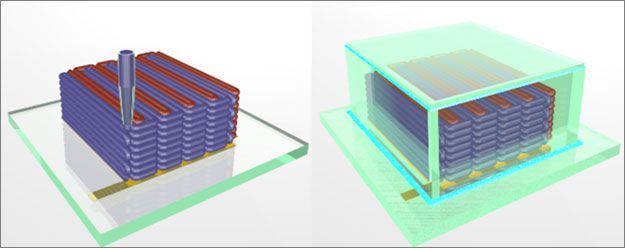
To create the microbattery, a custom-built 3D printer extrudes special
inks through a nozzle narrower than a human hair. Those inks solidify to
create the battery's anode (red) and cathode (purple), layer by layer. A
case (green) then encloses the electrodes and the electrolyte solution
added to create a working microbattery. [Credit: Ke Sun, Bok Yeop Ahn,
Jennifer Lewis, Shen J. Dillon]Wyss Institute
In recent years engineers have invented many miniaturized devices,
including medical implants, flying insect-like robots, and tiny cameras
and microphones that fit on a pair of glasses. But often the batteries
that power them are as large or larger than the devices themselves --
which defeats the purpose of building small.
To get around this problem, manufacturers have traditionally
deposited thin films of solid materials to build the electrodes.
However, due to their ultrathin design, these solid-state
micro-batteries do not pack sufficient energy to power tomorrow's
miniaturized devices.
The scientists realized they could pack more energy if they could
create stacks of tightly interlaced, ultrathin electrodes that were
built out of plane. For this they turned to 3D printing. 3D printers
follow instructions from three-dimensional computer drawings, depositing
successive layers of material -- inks -- to build a physical object
from the ground up, much like stacking a deck of cards one at a time.
The technique is used in a range of fields, from producing crowns in
dental labs to rapid prototyping of aerospace, automotive, and consumer
goods. Lewis' group has greatly expanded the capabilities of 3D
printing. They have designed a broad range of functional inks -- inks
with useful chemical and electrical properties. And they have used those
inks with their custom-built 3D printers to create precise structures
with the electronic, optical, mechanical, or biologically relevant
properties they want.
To print 3D electrodes, Lewis' group first created and tested several
specialized inks. Unlike the ink in an office inkjet printer, which
comes out as droplets of liquid that wet the page, the inks developed
for extrusion-based 3D printing must fulfill two difficult requirements.
They must exit fine nozzles like toothpaste from a tube, and they must
immediately harden into their final form.
In this case, the inks also had to function as electrochemically
active materials to create working anodes and cathodes, and they had to
harden into layers that are as narrow as those produced by thin-film
manufacturing methods. To accomplish these goals, the researchers
created an ink for the anode with nanoparticles of one lithium metal
oxide compound, and an ink for the cathode from nanoparticles of
another. The printer deposited the inks onto the teeth of two gold
combs, creating a tightly interlaced stack of anodes and cathodes. Then
the researchers packaged the electrodes into a tiny container and filled
it with an electrolyte solution to complete the battery.
Next, they measured how much energy could be packed into the tiny batteries, how much power they could deliver, and how long they held a charge. "The electrochemical performance is comparable to commercial batteries in terms of charge and discharge rate, cycle life and energy densities. We're just able to achieve this on a much smaller scale," Dillon said.
"Jennifer's innovative microbattery ink designs dramatically expand
the practical uses of 3D printing, and simultaneously open up entirely
new possibilities for miniaturization of all types of devices, both
medical and non-medical. It's tremendously exciting," said Wyss Founding
Director Donald Ingber, M.D., Ph.D.
Source: Wyss Institute