HealthManagement, Volume 17 - Issue 2, 2017
How can affordable cost 3D printed kidney phantoms assist nuclear medicine treatment planning and radiation dose optimisation?
Please briefly describe your research on 3D kidney phantoms.
Why did you set up this study and what were the main findings?
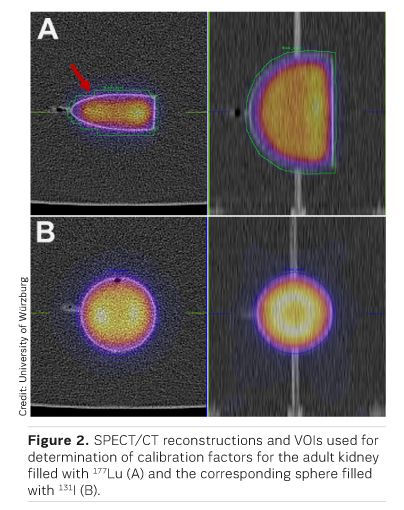
Nuclear medicine is a medical specialty that uses radiopharmaceuticals for diagnosis and treatment of disease. In the last years, driven by major advances in the biological understanding of metabolic processes, new radioactive compounds, particularly for cancer treatment, have been developed and applied preclinically and clinically. In many therapies involving peptides or small molecules (e.g. 177Lu-DOTATE, 177Lu- PS MA, 177Lu-Pentixather), the kidneys receive high absorbed doses and are considered the main organ at risk. Therefore, it is of special interest to perform accurate pre- and peritherapeutic kidney dosimetry, for which quantitative imaging forms the basis. Due to the lack or high cost of commercially available anthropomorphic phantoms, the aim of this research was to fabricate such phantoms using commercially available low-budget 3D printing hardware. For this purpose, a set of one-compartment kidney phantoms (newborn, 1-year-old, 5-year-old, adult) was developed. With this set of kidneys, an isotope-specific calibration for a commercial SPECT-CT gamma camera was performed. Although the presented one-compartment kidney models are perceived as an important step in the right direction, they show no significant calibration improvements over the typically used spherical phantoms, suggesting the necessity of more sophisticated, multi-compartment kidney models for achieving considerable improvements.
See Also: Sestamibi SPECT/CT Scan Improves Kidney Cancer Diagnosis
What did you find to be the main advantages of 3D printed kidney phantoms compared to conventionally produced phantoms?
All currently available imaging modalities for quantitative molecular imaging (PET-CT, SPECT-CT) suffer from a spatial resolution in the range of a few millimetres (PET) to centimetres (SPECT), which in comparison to other not inherently molecular imaging methods such as CT or MRI , leads to enhanced blurring of imaged anatomical structures, in turn impeding any quantification. By calibrating these imaging systems based on the activity distribution in 3D printed models, this insufficient resolution could be effectively recovered: the more realistic the model, the more accurate the process.

What are the next steps based on these results? Are you planning to develop more complex kidney phantoms or phantoms for other organs?
The aim of this study was to initially show the feasibility of low-budget 3D printing methods for the design and fabrication of anthropomorphic phantoms. Due to the simplification of the complex kidney anatomy to only one fillable compartment, however, only negligibly small differences to spherical phantoms—the standard in quantitative molecular imaging—were observed. Therefore, our next step will be the design and fabrication of multi-compartment kidneys, eg, consisting of an inner compartment resembling the renal medulla and pelvis as well as an outer compartment resembling the renal cortex, both of which can be filled separately.
What is needed to make this technique widely available so that other nuclear medicine departments can create individualised phantoms?
At this stage, the dimensions of the kidney designs are based
on a Medical Internal Radiation Dose committee (MIRD) recommendation—an organ
model widely accepted in the field of nuclear medicine. In addition to this
model, interested departments would need software for generating STereoLithography (STL) files of the desired model as well as 3D printing hardware
to produce the phantoms. Alternatively, the 3D printing could be outsourced to
a commercial service.
To standardise calibration of SPECT-CT systems, we are currently working on a set of multi-compartment kidney models which could be made available to interested institutions. Additionally, we are working on a standardised calibration scheme that, in combination with the set of phantoms, could pave the way to more standardised quantitative molecular imaging for dosimetry in molecular radiotherapies.
















