Vasoactive medication is one of the cornerstones in the treatment of critically ill patients in shock. Shock can be defined as a failure of the circulatory system to provide adequate tissue perfusion resulting in cellular injury and organ failure. The definitive treatment of any type of shock is treatment of the underlying disease and in the case of sepsis achieving source control. This means that during treatment of critically ill patients, use of vasoactive drugs is part of a multi-approach and complicated but coherent and concerted treatment plan. The most important contribution of vasoactive medication is the help for immediate restoration of sufficient cardiovascular circulation to buy time for further treatment. This means that even if you give the best possible vasoactive drug, the patient will still die if the rest of the treatment is insufficient. Part of the other treatment in critically ill patients consists generally speaking of fluid resuscitation, giving the right antibiotic at the right dose, mechanical ventilation, and specific underlying disease-related treatment that can be medical and/or surgical. Other issues such as specific nursing care, decubitus prevention, feeding, early start of activity, timely weaning of the ventilator, prevention of errors and complications, doing all the things right, etc. play an undeniable role in patient survival.
Vasoactive medication is generally discussed and studied apart from other elements of treatment and the effect on the cardiovascular circulation is considered pivotal. This is in fact odd, since the (short-term) cardiovascular circulatory status will depend on many other factors, such as fluid resuscitation and importantly, mechanical ventilation. Another issue is the significant inter-individual variation in terms of desirable cardiovascular parameters, which can also change for one individual over the short and long term. In general in published studies haemodynamic goals are given in terms of specific figures. Now where a specific figure of blood pressure at one moment may be good in one patient, it might be too low for another patient (e.g. in case of pre-existent hypertension) and higher than needed (for good organ function) for another patient. And since all vasoactive drugs have side-effects that are generally dose-dependent, if a patient receives a higher dose than required, the patient is more exposed to side-effects than the benefit of the given drug would justify. Therefore we introduce the goal of “enough” for different cardiovascular parameters. Enough is defined as: not too low and not too high for a specific patient. For example, the best cardiac output for a patient is “enough,” the best blood pressure and heart rate is also “enough.” The only missing link is then to define “enough” for each particular patient. Thinking this way would avoid designing a study where a mean blood pressure of 70 mmHg would be compared with 80 mmHg, or a study where different doses of a vasoactive drug are given to establish prefixed haemodynamic parameters to patients in order to compare the vasoactive drugs (DeBacker et al. 2010; Russell et al. 2008; Asfar et al. 2014). As will be explained this approach will also hold for the desirable amount of fluid resuscitation: enough. In order to be able to estimate at the bedside what is enough for a particular patient, knowledge and understanding of basic physiology and pharmacology is required.
Basic issues
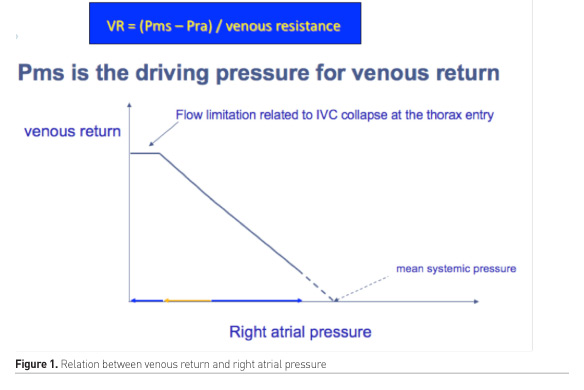
The pump generating the circulation of blood is placed in the thorax, where pressures vary according to the respiratory cycle, and the pump, a pressure chamber, is therefore placed in another pressure chamber. In normal conditions with spontaneous ventilation the intrathoracic pressure will lower during inspiration and return to normal pressure, which is slightly less than the atmospheric pressure. However, during mechanical ventilation the intrathoracic pressure, referring to the intrapleural pressure, is increased during inspiration and lowers, but remains above zero depending on the amount of PEEP (positive end-expiratory pressure) during expiration. Since blood flow is from areas with a higher pressure to a lower pressure, the intrathoracic pressure determines the pressure gradient for blood streaming towards the heart. It is of note that the most important task of the heart is to pump and transfer the amount of blood that is presented at the right side of the heart. Therefore, cardiac output is determined by the flow that is presented at the heart, and the venous flow that enters the heart equals the cardiac output. Similarly, the amount of volume expelled by the right side of the heart—through the pulmonary circulation—equals the volume that is presented to the left side of the heart and is subsequently expelled by the left ventricle. Because of this it is important for the practising intensivist to acknowledge that—as put forward by Guyton—the venous return is determined by the pressure gradient between the peripheral veins and the right heart, i.e. the right atrium or CVP. In a formula, including the venous resistance:
VR = (Pms-Pra)/venous resistance
Where VR = venous return, Pms = mean systemic pressure, Pra = right atrial pressure (Figure 1).
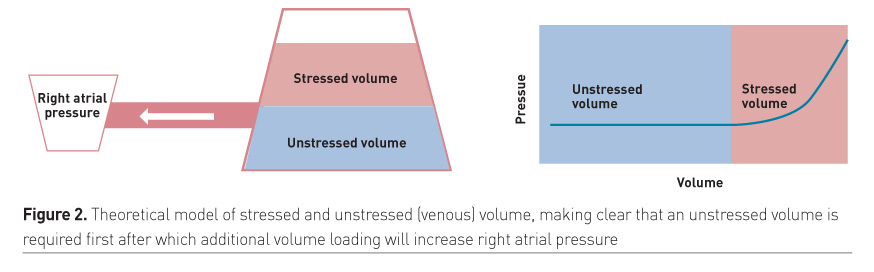
The venous system can be filled until the intravascular pressure starts to increase: this volume capacitance is called “unstressed volume.” With further filling the veins will be stretched and the intravascular pressure will increase: the capacitance of this further filling volume is called “stressed volume.” This theoretical model is depicted in Figure 2. When a fluid bolus increases the stressed volume, the driving force to the right atrium, right arterial pressure (RAP), can be increased, provided that the RAP will not increase accordingly. It is of note that with large volume resuscitation RAP may increase more than Pms, related to the reduced diastolic compliance of the heart and pericardium, resulting in a reduction of pressure difference Pms-Pra (Applegate et al. 1992).
Increasing intrathoracic pressure as seen in mechanical ventilation will reduce VR. On the other hand increased intrathoracic pressure will reduce afterload for the left ventricle. The effect of mechanical ventilation and lung volume on right ventricle afterload may vary according to the balance of stretching the extra-alveolar and intra-alveolar blood vessels: therefore increasing lung volume by e.g. PEEP may both lead to an increase and decrease of pulmonary vascular resistance (PVR) (Canada et al. 1982). Hyperinflation of the lung will result in overstretching of the alveolar vessels, thereby increasing PVR which can induce (acute) right ventricular failure. Hyperinflation can be the result of increased respiratory rate, larger tidal volumes (Vt) and insufficient expiratory time. Other heart-lung interactions, such as mechanical effects on the heart, and specific effects of sepsis on the heart and circulation are beyond the scope of this paper and can be read elsewhere (Marik and Bellomo 2016; Pinsky 2016; Fessler 1997). The situation and properties of the heart may even be more particular in case of previous coronary or hypertensive disease, with a huge spectrum of changes and severity.
In this context fluid responsiveness is frequently mentioned and an increase in stroke volume (SV) of 10-15% after a fluid challenge is considered as fluid responsiveness. The increase of myocardial contraction as a result of the stretching of actin and myosin muscular filaments (mostly indicated as according to the Frank-Starling principle) is in terms of energy consumption and demands a very favourable response of the heart. However, if the heart does not respond with an increase of SV to a fluid load, the latter might be harmful due to induction of oedema, which in turn may cause all kind of unfavourable effects for diverse organ functions (Hilton and Bellomo 2012). A recent meta-analysis of haemodynamically unstable patients showed that 50% remain fluid responsive after the initial resuscitation (Bentzer et al. 2016). Predictors for a positive fluid response are the passive leg raise test and pulse pressure variation (Bentzer et al. 2016). In terms of clinical parameters this has been translated in pulmonary and peripheral oedema, abdominal compartment syndrome, kidney injury and longer mechanical ventilation (Malbrain et al. 2014). Not surprisingly, there is an increasing focus on potential adverse effects caused by (too much) intravenous fluid resuscitation and this underscores that one should give just enough to a patient. This perception has contributed to a shift of earlier use of vasoactive drugs to obtain haemodynamic goals in patients and to use less fluids.
From the previous it may become clear that execution of mechanical ventilation and fluid resuscitation, timely treatment of underlying disease as well as the intrinsic properties of the heart at a particular moment will influence the effect of any vasoactive drug. It is therefore extremely surprising that trials evaluating vasoactive drugs do not take mechanical ventilation and specific properties that are known to be of importance, into account.
Markers of “enough”
Although many studies, including randomised clinical trials (RCTs), focus on a single parameter (e.g. plasma lactate levels, SvO2, cardiac output), clinicians use several parameters simultaneously to assess the situation and possible improvement of the condition of a patient. These parameters include signs of adequate organ function and perfusion: mental state, peripheral skin perfusion as determined by nose or knee temperature and mottled skin, capillary refill, diuresis, SvO2, lactate levels, oxygenation, blood pressure, ECG abnormalities, venous curves on the monitor, cardiac output if measured. The experienced clinician will very much focus on pattern recognition of all these simultaneously assessed parameters. The response to any intervention will help to estimate whether the chosen therapy is favourable or not. The experienced clinician will furthermore use all available data and will not be guided by one single parameter alone, e.g. focus only on cardiac output. Errors as made in the past, such as striving for “supranormal” oxygen delivery (DO2) with excessive fluid administration and very high doses of inotropes or very strict regulation of serum glucose levels with very high doses of insulin and at the price of causing hypoglycaemia, should now turn the intensivist into a physician who realises that just enough is enough and better is the enemy of good (Voltaire). Translated in an example: if a patient is hypotensive, anuric and confused, and responds to two times 250 mL of Ringers lactate intravenously with an increase in blood pressure, a lowering of heart rate and improved mental state and diuresis, the amount of given fluid was enough for this moment. Blood lactate levels may also be helpful to assess clinical deterioration or improvement of a patient over time, but one should realise that lactate can be increased as a result of anaerobic glycolysis due to systemic or regional hypoperfusion, but also due to stress-related adrenergic-induced aerobic glycolysis, impaired hepatic clearance as well as mitochondrial dysfunction limiting pyruvate metabolism. One should therefore never focus on a single parameter such as lactate alone to assess the effect of treatment and there is insufficient convincing evidence that decreasing lactate levels alone is a useful target of therapy in critically ill patients (Bakker 2014).
Goal for blood pressure: enough
For the treatment of shock, blood pressure has been and is a pivotal marker of severity of shock and effectivity of treatment. As outlined before, we argue that blood pressure should be just enough. Now it is clear that “enough” is different and a higher value in a patient with shock and a history of insufficiently treated hypertension compared to a person who is used to a blood pressure of 105/70 mmHg and has a blank medical history. Furthermore, since all treatments involved such as fluid resuscitation and the administration of vasoactive drugs are known to produce important side-effects such as oedema and arrhythmias, which are also dose-dependent, it becomes clear that the lowest possible dose/quantity should be given: the definition of “enough”. Only in this way will the advantages outweigh the disadvantages for the patient. We have therefore reasons to believe that RCTs on vasoactive drugs, aiming at specific figures, or even comparing different figures of blood pressure, lack sufficient acknowledgement of physiological facts. The recommendations of the Surviving Sepsis Campaign to maintain a mean arterial pressure (MAP) of 65 mmHg in septic shock may be of use for paramedics and inexperienced physicians, but as follows from the previous text should not be used by experienced intensivists who are aware of the cardiovascular physiology and heart-lung interactions.
However, things are not that simple. The blood pressure that is enough for the brain might not be enough for the kidneys or vice versa. Some organs such as the brain, heart and kidney are known to have some ability to auto-regulate blood flow resulting in a constant blood flow across a specific MAP range (Hollenberg 2011). But this can be deranged in different situations, such as brain injury, pre-existing hypertension or abdominal compartment syndrome. Therefore it is of utmost importance to evaluate clinical signs and markers of tissue perfusion continuously. Measurement of cardiac output can be complementary to understand the present (circulatory) physiology of the patient, either done by thermodilution with a pulmonary artery catheter, or combined with continuous pulse contour analysis through an arterial cannula, or noninvasive using cardiac ultrasound or pulse contour analysis. Cardiac output should not be aimed at a specific target, but again should be “enough” and certainly not too high, as has been previously advised for supra-normal goals of therapy.
Use of vasoactive drugs
In clinical practice, to determine which vasoactive drug to administer, the desired effect has to be determined as well as knowledge about the required receptors or other cellular pathways involved. Unfortunately, there is not such a drug that 100% specifically stimulates only one type of receptor. And most adrenergic drugs have a combined effect on both α- and β-adrenergic receptors. To increase arterial blood pressure an adrenergic drug that has a predominant α1-adrenergic effect is required, or a drug that stimulates another receptor or pathway such as angiotensin II or vasopressin. In cardiogenic shock, low cardiac output state or other types of impaired cardiac function (where cardiac output is “not enough”), an agent may be required that has affinity for β1-adrenergic receptors to increase inotropy, chronotropy or dromotropy. There are also other cellular pathways to increase the cardiac contractility via other routes, such as increasing intracellular calcium or affinity of cardiac myocytes for calcium. It is good to realise that any drug has potential adverse effects. Vasopressor therapy can result in decreased stroke volume and thus cardiac output because of the increased afterload.
During inotrope use arrhythmias might occur, and cardiac oxygen demand might be increased because of increased heart rate. Inotropes can also cause vasodilatation requiring additional vasopressors to maintain adequate blood pressure. In general, short-acting vasoactive drugs should be titrated to effect to achieve specific haemodynamic goals while minimising potential harmful effects.
We provide a brief overview of the pharmacotherapy with vasoactive drugs in critical care medicine in an attempt to summarise cellular effects, indications, common adverse effects as well as recent scientific evidence, for both proven drugs and newer agents.
Adrenaline (Epinephrine)
Adrenaline is an endogenous hormone and neurotransmitter produced by and stored in the adrenal glands. It has a main effect on the β1 receptor, with additional affinity for β2- and α1- adrenergic receptors, resulting in both increased cardiac output and mean arterial pressure. The main difference beween adrenaline and noradrenaline (norepinephrine) is the increased affinity of adrenaline for β2-receptors compared to noradrenaline. Low doses of adrenaline result in an increase of cardiac output and variable effects on mean arterial pressure, depending on the balance of effects of β1, β2 and α1 adrenergic receptors stimulation.
It is short acting and mostly metabolised in the liver by catechol-O-methyltransferase (COMT) and monoamine oxidases (MAO). Excretion of metabolites is renal. Because of strong β1 effect, arrhythmias are common during use. Lactic acidosis is often reported, which is a direct β2 adrenergic effect.
Current indications for the use of adrenaline in critically ill patients are during cardiac arrest, anaphylactic shock and as an adjunctive antihypotensive agent. There are few trials that compare adrenaline to other agents, of which two larger studies can be mentioned. One randomised controlled trial (RCT) (n=280) compared adrenaline to noradrenaline in septic shock and found no difference in survival but a higher incidence of tachyarrhythmia and lactic acidosis in the adrenaline group (Myburgh et al. 2008). Another RCT (n=330) compared adrenaline and a noradrenaline/dobutamine combination and concluded there was no difference in safety and efficacy (Annane et al. 2007).
In cardiopulmonary resuscitation adrenaline appears to increase the chances for return of spontaneous circulation, but does not increase favourable neurological outcome. A very recent placebo-controlled RCT confirmed these findings, with adrenaline improving 30-day survival, but no difference in proportion of patients surviving hospital discharge with a favourable neurological outcome (Perkins et al. 2018).
Noradrenaline
Noradrenaline is also an endogenous hormone and neurotransmitter. Its main affinity is for α1-adrenergic receptors, with some β1 and minor β2 effects. Noradrenaline has a short half-life; it is active for about 1-2 minutes. Metabolism is hepatic and in nerve endings and inactive metabolites are excreted renally. Noradrenaline is used mainly as a vasopressor in vasodilatory shock, but also cardiogenic shock and during general anaesthesia for anaesthesia-induced hypotension. Adverse effects are mainly related to severe vasoconstriction, such as limb or gastrointestinal ischaemia. A widespread misunderstanding is that noradrenaline decreases coronary blood flow (Mueller et al. 1970). The comparison of noradrenaline versus dopamine in the treatment of septic shock seemed more favourable towards noradrenaline looking at clinical trials (De Backer et al. 2012)
Dopamine
Dopamine is the endogenous precursor of (nor)adrenaline and has a complicated action on the cardiovascular, renal and neurohumoural systems. Metabolism is fast, elimination half-life is 1-2 minutes with metabolism in the liver, kidneys and plasma by MAO and COMT into inactive metabolites and noradrenaline. Metabolites are excreted by the kidneys. The circulatory effect depends on the predominant effect of the different receptors that are stimulated by dopamine: dopaminergic receptors (DA1, DA2), β1- and α1-adrenergic receptors (Girbes et al. 2000). Some authors wrongly still write that dopamine stimulates different receptors at different doses, distinguishing 1-4 µg/kg/min, 4-10 and >10 µg/kg/min. This is simply not true. At every dose dopamine stimulates DA1, DA2, β1- (β2), and α1-adrenergic receptors. Additionally, dopamine inhibits uptake-1. In the past the positive effects of dopamine were overrated, mainly related to its effects on renal blood flow (Girbes and Smit, 1997). Dopamine was given at “a renal dose”, i.e. a dose up to 4 µg/kg/min to preserve renal function, but later studies indicated no beneficial effects in the long-term (Marik 2002). And since the RCTs comparing noradrenaline and dopamine, the use of dopamine is significantly reduced, due to its reported side-effects, mainly dysrhythmias.
Dobutamine
Dobutamine is a mixture of two isomers with mainly β1, but also β2 and mild α1-adrenergic receptor effects. Elimination half-life is 2 minutes, metabolism is mostly by COMT in the liver and tissues into inactive metabolites that are excreted by the kidney. The main effect of dobutamine is as an inotrope through β1-receptor effects: an increase in cardiac output by increasing stroke volume and heart rate. Effects on blood pressure vary and are unpredictable (Hollenberg 2011). Because of the positive chronotropic and inotropic effect on the heart, dobutamine causes an increase in myocardial oxygen demand, with a risk of myocardial ischaemia. Ventricular arrhythmia may also occur.
Dobutamine is an agent widely used in cardiogenic shock, and gained popularity as an agent used in septic shock since the original early goal-directed therapy (EGDT) trial where it was used to achieve central venous oxygen saturation goals (Rivers et al. 2001). It was only realised later by intensivists that the Rivers study population was not a representative population of patients with sepsis. Many other large studies proved no benefit for the use of dobutamine as deemed indicated according to the EGDT guidelines (Mouncey et al. 2015).
Vasopressin and analogues
Antidiuretic hormone, also named arginine vasopressin (AVP), is an endogenous hormone secreted by the anterior pituitary gland in response to stress or shock. There are three major vasopressin receptors and for conditions of circulatory shock the vasoconstrictor effect of smooth muscle caused by activation of the V1 receptor is most relevant. AVP is given intravenously and has a half-life of 10-35 minutes with V1, V2 and V3-receptor affinity. It is rapidly metabolised by the liver and kidney.
Terlipressin is a non-selective vasopressin analogue that also is a prodrug of lysine vasopressin, which is similar to AVP. The elimination half-life is 50 minutes, with active metabolites for up to 6 hours. Vasoconstriction caused by V1 receptor stimulation could lead to additional cardiac ischaemia and is possibly more pronounced in the mesenterial region resulting in gastrointestinal ischaemia. Reflex bradycardia may lead to decreased cardiac output. V2-receptor activation causes endothelial von Willebrand factor release, causing enhanced platelet aggregation with increased risk for thrombosis. (Saad and Maybauer 2017).
Selepressin is a novel, short-acting selective V1 agonist that appears promising in initial studies, but a critical appraisal of new data must be awaited. In animal sepsis models, improved survival compared to noradrenaline is reported. And importantly, a decrease in pulmonary capillary leak (Saad 2017).
There is no convincing evidence that vasopressin or its analogues are superior to catecholamines in the treatment of sepsis or during cardiopulmonary resuscitation. Vasopressin can however be considered as a second-line vasopressor therapy. The vasopressin analogue terlipressin is commonly used in hepatorenal syndrome and portal hypertension but there is no convincing evidence supporting its use in septic shock. The clinical relevance of studies showing that vasopressin reduces the use of noradrenaline are in our view questionable.
Angiotensin 2
Synthetic (or bovine) angiotensin II is a novel drug that is postulated as a third type of vasopressor, after adrenergic vasopressors and vasopressin. Angiotensin II is converted from angiotensin I by angiotensin-converting enzyme (ACE) and a product of the renin-angiotensin-aldosterone system (RAAS), which is activated by decreased renal perfusion in hypovolaemia. Angiotensin II has a vasoconstrictor effect by increasing intracellular calcium levels in smooth muscle cells that results in vascular contraction after activating several cell-signaling pathways. There is however, little evidence supporting the use of angiotensin II in shocked critically ill patients. The recent Angiotensin II for the Treatment of High-Output Shock (ATHOS-3) trial investigating the addition of angiotensin II to noradrenaline in the treatment of “refractory” vasodilatory shock in 344 patients showed a significant increase in blood pressure and decrease of noradrenaline dose in the study group. However, there is a lack of data on serum lactate or central venous oxygen saturation and, most importantly, none of the patients was in fact treated with high-doses vasopressor therapy at the beginning of the study. There were no significant differences in mortality or adverse events (Khanna et al. 2017).
Nitric Oxide (NO) inhibitors
Systemic inflammation can cause the overexpression of “inducible nitric oxide synthetase” (iNOS), which is stimulated by pro-inflammatory cytokines, resulting in excess nitric oxide and vasodilatation. Attempts have been made to counteract this overexpression. However, clinical trials have been disappointing and at this moment there is no place for the use of NO-inhibitors in the treatment of septic critically ill patients.
Phosphodiesterase inhibitors: milrinone and enoximone
Frequently used phosphodiesterase inhibitors (PDEi) in the ICU are enoximone and milrinone. Both inhibit phosphodiesterase type III, resulting in an increased amount of intracellular cAMP, which leads to an activation of cardiac calcium channels and increased calcium influx during systole increasing cardiac contraction force. During diastole, there is an increased efflux of calcium increasing relaxation (lusitropy). Because of the inotropic and vasodilatory effects, PDEi are also called inodilators. Excretion is mostly renal, and half-life depends greatly on kidney function, with the risk of accumulation. Known adverse events include tachyarrhythmia, thrombocytopaenia and hypotension. Although many studies found an improvement of circulatory parameters in patients with (severe) heart failure in the short term, later studies showed that this was not translated into a better long-term outcome. For patients after cardiac surgery that require inotropic support, there is also a possible trend towards greater mortality compared to dobutamine (Nielsen et al. 2018).
Levosimendan
Levosimendan is a calcium sensitiser: it increases the susceptibility of cardiac myocytes to calcium by binding on troponin C, resulting in increased cardiac contraction without increasing calcium levels. There is no change in diastolic relaxation or increase in myocardial oxygen demand. Another effect is vasodilatation by opening vascular smooth muscle cells as a result of opening ATP-sensitive potassium channels. The biological half-life is about 1 hour and active metabolites after conjugation with glutathione, the metabolites are pharmacologically inactive and excreted in urine and faeces. Some metabolites, 4-7% of the levosimendan dose, are formed slowly and the elimation half-life has been up to 70-80 hours in patients with congestive heart failure. A single day of infusion is adequate for several days of treatment. It is therefore not surprising that the use of levosimendan has been associated with higher incidence of tachyarrhythmia and prolonged hypotension (Antila et al. 2007). Initial enthusiasm about the application of levosimendan for either low cardiac output states or sepsis has been denied by later and larger studies.
Other agents
Phenylephrine is mainly an α1-agonist, that is used mainly in theatre for perioperative hypotension. Because of the increase in systemic vascular resistance (SVR) and subsequent baroreceptor-induced decrease in heart rate, its net effect is a decrease in cardiac output. Ephedrine is an indirect agent that stimulates the release of endogenous noradrenaline. Repeated doses show decreased effect known as tachyphylaxis, caused by a depletion of cellular noradrenaline stores. Data on patients in the ICU are scarce and there are no data to justify prolonged administration in the ICU. Isoprenaline is a short-acting nonselective beta agonist that is mostly used in treatment of extreme bradycardia and atrioventricular block.
Special considerations
Pulmonary arterial hypertension
Pulmonary arterial hypertension (PAH) has obtained more attention in the ICU with a focus on right ventricular (RV) function. In critically ill patients with pulmonary hypertension and (imminent) RV failure, the goal of treatment is to reduce pulmonary vascular resistance, improve right ventricular contractility, optimising right ventricular preload while also maintaining adequate systemic perfusion. Fluid management requires special attention since in the case of increased RV afterload, volume loading will result in RV dilatation and displacement of the interventricular septum toward the left ventricle (LV) with impaired LV diastolic filling as well as decreased right coronary perfusion. Failure to take this into account will thus result in deterioration of the circulation with deleterious consequences. The most important factor is to establish the diagnosis as soon as possible in order to be able to provide adjusted precision therapy. Cautious fluid resuscitation (including no extra fluid administration) is an important factor for that and the reader is referred to a recent review (Jentzer and Mathier 2016). The goals for vasoactive medication include reduction of PVR, maintenance of SVR and increased cardiac output. PDEi inhibitors have favourable effects compared to dopamine and dobutamine as a result of increased right ventricle contractility and pulmonary vasodilatation with less tachycardia and additional oxygen consumption. The decrease of SVR and concomitant decrease of systemic blood pressure may be treated with a vasopressor such as noradrenaline. However, all α-1 agonists are reported to increase PVR, with a possible increase in right ventricular afterload. Vasopressin has a moderate additional and considered favourable effect of endothelin-dependent pulmonary vasodilation, thereby producing less increase of PVR compared to the increase of SVR. Intravenous prostanoids are indicated for patients with critical PAH, with a preference for prostanoids with a short half-life e.g. epoprostenol.
Hepatic failure
Patients with hepatic failure are often volume depleted as a result of low systemic vascular resistance (vasodilation), together with a high cardiac output, and it mimics vasodilatory shock. Few studies have been conducted comparing different vasoactive drugs in the critically ill with hepatic failure, and most have very few patients. After volume resuscitation, noradrenaline is usually recommended because of its less outspoken constrictor effect on the splanchnic circulation. Vasopressin or terlipressin can be added to potentiate noradrenaline. There is insufficient reason to believe that terlipressin is superior to other vasoactive drugs in case of hepatorenal syndrome (Israelsen et al. 2017).
Traumatic brain injury
In physiological conditions, the brain has the ability to auto-regulate cerebral blood flow. After TBI, cerebral autoregulation could be impaired, and therefore a pivotal treatment goal in the critical care setting is to maintain adequate cerebral perfusion. A commonly used parameter to guide treatment is cerebral perfusion pressure, which is the function of mean arterial pressure minus intracranial pressure measured by an intraventricular or intraparenchymal probe. The recommended cerebral perfusion pressure is between 60-70 mmHg (Carney et al. 2016). There are no high-quality studies indicating which vasoactive drug is best used to increase arterial pressure in TBI patients. Noradrenaline has the most predictable effects.
Discussion
Treatment of critically ill patients is micromanagement of all vital functions. The use of vasoactive medication is only a part of the treatment of a complex pathophysiology. Because of the immense heterogeneity amongst patients, the increasing choice between different agents and the supportive nature of treatment with vasoactive drugs, comparing treatments is challenging and prone to error. Although we acknowledge the huge efforts and organisational skills to perform RCTs with vasoactive drugs, we question the value in terms of external validation and applicability, especially in view of individual precision medicine, for the reasons mentioned above. The value of such trials is therefore in our opinion merely on the collection of data of side-effects. Furthermore, one should realise that if such trials compare different strategies of the use of vasoactive drugs, the outcome applies only for that specific strategy with those defined haemodynamic goals. In other words, if a trial says that noradrenaline is “better” than dopamine, it is only applicable if you use the specific haemodynamic goals in a similar population. And the result might be different if you use a slightly different haemodynamic goal for dopamine or noradrenaline. We also foresee that the answer on which is the best vasoactive medication for my patient, or groups of patients, will never come from large RCTs. Recommendations with fixed figures such as in the Surviving Sepsis Campaign are perhaps of use for non-intensivists, but are potentially dangerous and ignore the complexity of disease in individual critically ill patients. Therefore, understanding the pathophysiology, watching carefully and continuously the effect of the given therapy, doing all (simple) things right will for the coming decades remain the cornerstone of therapy with vasoactive medication, but it will be called precision medicine.
Conflict of interest
Jon Gutteling declares that he has no conflict of interest. Armand R.J. Girbes declares that he has no conflict of interest.