ICU Management & Practice, Volume 21 - Issue 6, 2021
Introduction
Critical illness is common in patients with haematologic malignancies. Recent data demonstrate that 14% of patients will require ICU admission within one year of their initial diagnosis, with the highest risk in acute leukaemia (Ferreyro et al. 2021). As cancer care, infectious disease practices and critical care management have evolved, ICU outcomes in this population have improved. Furthermore, critical illness is increasingly being recognised as part of the treatment pathway for a subset of patients receiving intensive therapies to achieve cure or sustained disease control. Given this, the current and future states of Oncologic Critical Care management for this population will require increased collaboration between intensivists and haemato-oncologists. While sepsis and respiratory failure are the most common admitting diagnoses, there are a range of disease- and treatment-specific complications that intensivists must be aware of to provide optimal care for these complex patients. This review will provide an overview of emergencies that are commonly seen in malignant haematology including hyperleukocytosis, leukostasis, tumour lysis syndrome, disseminated intravascular coagulation, neutropenic sepsis, hyperviscosity syndrome, side effects of novel therapies, and complications of allogeneic haematologic stem cell transplant.
Epidemiology
The modern 1-year incidence of ICU admission after diagnosis of haematologic malignancies is 14% ranging from 7% for indolent lymphoma to 23% for acute myeloid leukaemia (AML) followed by aggressive non-Hodgkin’s lymphoma (18%) (Ferreyro et al. 2021). Half of this cohort are admitted within 30 days of diagnosis and the median time from diagnosis to admission in a large population based cohort study is 35 days (Ferreyro et al. 2021). As novel therapies increase as part of the standard of care, an increase in the incidence of critical illness will likely be seen.
Acute Myeloid Leukaemia with Hyperleukocytosis
Up to 20% of patients with acute myeloid leukaemia (AML) will present with an extreme elevation in their white blood cell count (WBC) known as hyperleukocytosis (Rollig et al. 2015; Ali et al. 2016). Generally defined by a WBC > 50-100 x 109/L, hyperleukocytosis is a medical emergency. Without rapid recognition and treatment, early mortality rates are high, with intracranial haemorrhage and respiratory failure as frequent causes of death (Marbello et al. 2008). Cytoreduction is the definitive treatment but excellent supportive care and prompt recognition of complications are essential. For patients who do not receive intensive chemotherapy, the mortality rate may exceed 50% at 30 days (Shallis et al. 2020).
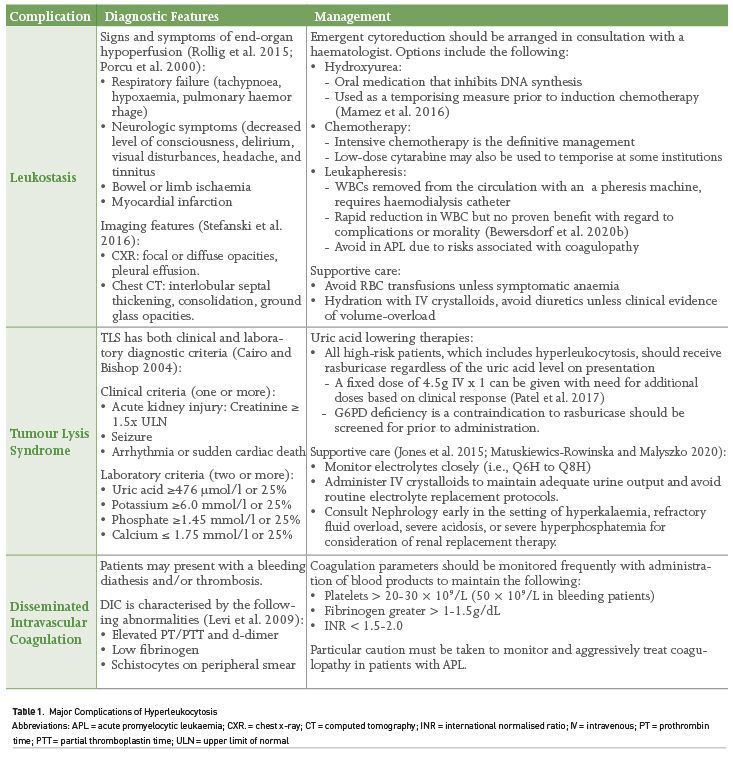
The most concerning complications of hyperleukocytosis include leukostasis, tumour lysis syndrome (TLS) and disseminated intravascular coagulation (DIC) with the diagnostic features and management considerations outlined in Table 1. Leukostasis is a clinical diagnosis based on evidence of end-organ hypoperfusion in a patient with hyperleukocytosis. It results from occlusion of the microcirculation with endothelial damage and is much more common in AML than ALL due to the large size of the myeloid blasts (Porcu et al. 2000). Respiratory or neurologic symptoms are the most common clinical manifestations of leukostasis and may be the reason for ICU admission (Stahl et al. 2020).
Tumour Lysis Syndrome
Tumour lysis syndrome (TLS) results from the rapid breakdown of cancer cells and release of intracellular contents. It can occur either spontaneously or after initiation of cytoreductive therapies and may present clinically as acute kidney injury (AKI), arrhythmias, seizures, or sudden death. TLS can present in 25% of patients with AML and hyperleukocytosis, with similarly high incidence reported in the paediatric literature for ALL (Shallis et al. 2020; Stahl et al. 2020; Bewersdorf et al. 2020a; Truong et al. 2007). Patients with aggressive lymphomas or significant disease burden such as Burkitt lymphoma, lymphoblastic lymphoma, and DLBCL, T-cell lymphoma with elevated LDH, or bulky disease on CT are also at high risk (Jones et al. 2015). Risk for death from TLS is highest in AML, with mortality rates that may approach 30% for patients hospitalised (Durani et al. 2017).
The approach to diagnosis and management are outlined in Table 1. In addition to administering uric acid lowering therapies and addressing the sequelae of acute kidney injury, the primary management is supportive with frequent monitoring. All high-risk patients, or those that have already progressed to TLS, should receive rasburicase. Rasburicase is recombinant urate oxidase that leads to the breakdown of uric acid. This is in contrast to allopurinol which reduces uric acid formation and is appropriate when the TLS risk is low to moderate. IV fluids should be given to maintain adequate urine output, routine electrolyte replacement protocols should be avoided, and nephrology should be consulted for consideration of dialysis in the event of progressive AKI, acidosis, or electrolyte disturbances. Contrary to historical practices, there is no evidence for urinary alkalinisation, and diuretics should generally be avoided unless there is concern for volume overload.
Acute Promyelocytic Leukaemia
Acute promyelocytic leukaemia (APL) is a variant of AML that is seen in approximately 10-15% of cases (Tallman and Altman 2009). While APL is uncommon, it is important to be aware of it due to its unique complications and prognostic considerations. The molecular hallmark of APL is a translocation between chromosome 15 and 17 leading to the production of a the PML/RARα fusion gene. Clinically, patients present with cytopenia, coagulopathy, and bleeding and are considered to have high risk disease if the WBC is elevated (greater than 10x109/L). While the overall outcomes for APL are excellent, 20-25% of patients may die in the first 30 days largely due to haemorrhagic complications with a significant portion of these deaths in the first week (Lehmann et al. 2017; Micol et al. 2014). For patients who survive this initial period, long-term survival approaches 90%, underscoring the importance of excellent supportive care (Coombs et al. 2015; Yilmaz et al. 2021).
When APL is suspected, treatment with all-trans retinoic acid (ATRA) should be started emergently as per the treating haematologist even if the diagnosis is not confirmed. Treatments administered for APL in addition to ATRA include arsenic trioxide (ATO) and chemotherapy. The primary consideration for the intensivist is monitoring for and aggressively treating coagulopathy. Coagulation parameters should be checked frequently, and blood products should be administered to keep platelets greater than 30 × 109/L (50 × 109/L in bleeding patients), fibrinogen greater than 1-1.5g/dL, and INR less than 1.5 (Sanz et al. 2019). After treatment is initiated, approximately 25% of patients may develop ATRA differentiation syndrome which manifests as fever, hypotension, respiratory failure with interstitial infiltrates, AKI, pleural or pericardial effusions, and peripheral oedema (Montesinos et al. 2009). The treatment is dexamethasone 10mg IV Q12H which should be started at the first sign of any symptoms (Sanz et al. 2019). Prophylactic steroids may also be considered in patients being started on ATRA, particularly in the setting of an elevated WBC. This decision should be discussed with the malignant haematology team.
Febrile Neutropenia and Neutropenic Sepsis
Febrile neutropenia (FN) is defined as a temperature ≥ 38.3°C or a temperature ≥ 38.0°C lasting more than 1 hour with an absolute neutrophil count (ANC) < 0.5 × 109/L (Freifeld et al. 2011). Following induction chemotherapy, patients with acute leukaemia may develop profound, prolonged neutropenia with an ANC < 0.1 x 109/L lasting over 1 week. This places them at high risk for infection and neutropenic sepsis. Patients with leukaemia are at the highest risk of death from FN among all patients with cancer with invasive fungal infections, candidaemia, bacteraemia, pneumonia, and the presence of comorbidities associated with a further increase in mortality (Kuderer et al. 2006). While patients with lymphoma may develop FN, the degree and duration of neutropenia are typically less than that seen in acute leukaemia, decreasing the overall risk of severe infection. However, it is important to discuss the anticipated course based on the underlying malignancy and therapy received with the patient’s haemato-oncologist.
The core management principles for sepsis apply to neutropenic patients including early recognition, urgent administration of antibiotics, source control, fluid resuscitation as needed, and haemodynamic support to maintain end-organ perfusion (Kochanek et al. 2019). However, the diagnostic work-up to determine the source is frequently more extensive and empiric antibiotic coverage is broader due to different risk factors in the setting of profound immunosuppression (Figure 1). Bacteraemia from central lines, gut translocation of organisms, and pneumonia are common causes of infection in the setting of neutropenia. Granulocyte colony stimulating factor (GCS-F) may be considered as an adjunctive treatment to reduce the duration and severity of neutropenia and can be discussed with oncology (Mhaskar et al. 2014). However, there is also concern that GCS-F may precipitate or worsen respiratory failure during neutrophil recovery (Mignard et al. 2019).

One specific aetiology of FN that is important to consider in any patient with gastrointestinal symptoms is neutropenic enterocolitis (NE). The diagnosis is based on the presence of abdominal pain and bowel wall thickening of more than 4mm on CT in a patient with FN (Gorschluter et al. 2005). The initial management is supportive with bowel rest, fluid resuscitation, and broad-spectrum antibiotics. C. difficile infection should also be ruled out. Consideration should be given to empiric antifungal coverage as high rates of fungal infection have been reported in critically ill patients with NE, particularly when there is small bowel involvement on CT (Duceau et al. 2019). Surgical management is generally reserved for complications such as bowel perforation or necrosis, uncontrolled bleeding, or abscess formation (Rodrigues et al. 2017). Early surgery consultation is warranted in critically ill patients (Saillard et al. 2018).
Superior Vena Cava Syndrome
Patients with lymphoma and bulky mediastinal disease or lymph node involvement are at risk for developing superior vena cava (SVC) syndrome. Progressive SVC obstruction from exterior compression, infiltration, or thrombosis leads to decreased venous drainage into the right atrium and corresponding symptoms of increased venous pressure including oedema of the face and arms, dyspnoea, cough, and development of vascular collaterals in the upper chest. In severe cases, symptoms of airway or cerebral oedema may develop, and rarely, patients may have haemodynamic collapse. The best test to confirm the diagnosis is CT venogram, with MRI as another option (Friedman et al. 2017). However, care must be taken to ensure patients are able to safely lay flat as compressive symptoms may significantly worsen in the supine position.
The major considerations for the intensivist surround airway management and haemodynamic support. Due to the risk of airway collapse from mediastinal compression on induction of anaesthesia and airway oedema from venous engorgement, an airway expert should be involved during intubation. Awake fiberoptic intubation should be performed by an experienced provider with the head of the bed in an upright position (Chaudhary et al. 2012). To maintain preload, IV fluids should be given via lower extremity access with vasopressors as needed to achieve an adequate mean arterial pressure. In cases of severe airway compromise where intubation is not possible, extracorporeal life support (ECLS) as a bridge to therapy can be considered (Leow et al. 2021).
In the case of life-threatening symptoms, endovascular stenting is an effective first-line treatment associated with minimal complications (Lanciego et al. 2009). Catheter-directed thrombolysis can also be performed if thrombosis is contributing to the obstruction (Rachapalli and Boucher 2014). If the diagnosis is not previously known, urgent biopsy should also be arranged. However, if safe to do so, decisions regarding stenting should be deferred until a definitive management plan is discussed with the malignant haematology team as chemotherapy and/or radiation may also be effective therapy.
Hyperviscosity Syndrome
Hyperviscosity syndrome is an emergency that results from elevated levels of monoclonal protein in the blood and can be life-threatening if not promptly recognised and treated. It is most commonly seen in Waldenstrom’s macroglobulinaemia (WM) which is a rare subtype of non-Hodgkin lymphoma (NHL). In WM, there is abnormal production of monoclonal IgM protein leading to a rise in serum viscosity which is the primary indication for treatment in 17% of patients (Dimopoulos and Kastritis 2019). Hyperviscosity syndrome can also be seen in a small proportion of patients with multiple myeloma (Weaver et al. 2020).
Once serum viscosity rises above 4 centipoise with the presence of a monoclonal protein, patients may become symptomatic with mucocutaneous bleeding, neurologic symptoms, visual disturbances, fatigue, and generalised malaise (Crawford et al. 1985; Castillo et al. 2016). In severe cases, permanent vision loss, coma, and seizures may occur. Fundoscopic exam is help-fully diagnostically with the presence of dilated and tortuous veins, haemorrhages, papilledema, and exudates supportive of the diagnosis (Stone and Bogen 2012). If serum viscosity testing is not available, the immunoglobulin level can be used as a substitute. Concern for hyperviscosity is increased with IgM greater than 3 g/dL, IgG greater than 4 g/dL, or IgA greater 6 g/dL (Mehta and Singhal 2003).
Therapeutic plasma exchange (TPE) to reduce IgM levels is the primary treatment and should be instituted when clinical suspicion is high, even in the absence of serum viscosity testing. While this is being arranged, care should be taken to avoid any treatments that may increase plasma viscosity such as RBC transfusions. Plasmapheresis is generally continued to maintain serum viscosity below the level that results in symptoms, which may vary between individuals (Stone and Bogen 2012). Chemotherapy to treat the underlying disease is the definitive management. However, caution must be maintained in patients receiving rituximab for WM as it may result in an increase in IgM levels in a proportion of patients requiring consideration for pre-emptive TPE (Weaver et al. 2006).
Side-Effects of Novel Therapies
Treatment of haematologic malignancies is rapidly changing with increasing use of novel therapies which leverage the immune system to eliminate cancer cells. Two of these immuno-therapies: immune checkpoint inhibitors (ICIs) and chimeric antigen receptor (CAR) T-cells, may lead to severe and reversible side effects that can result in critical illness.
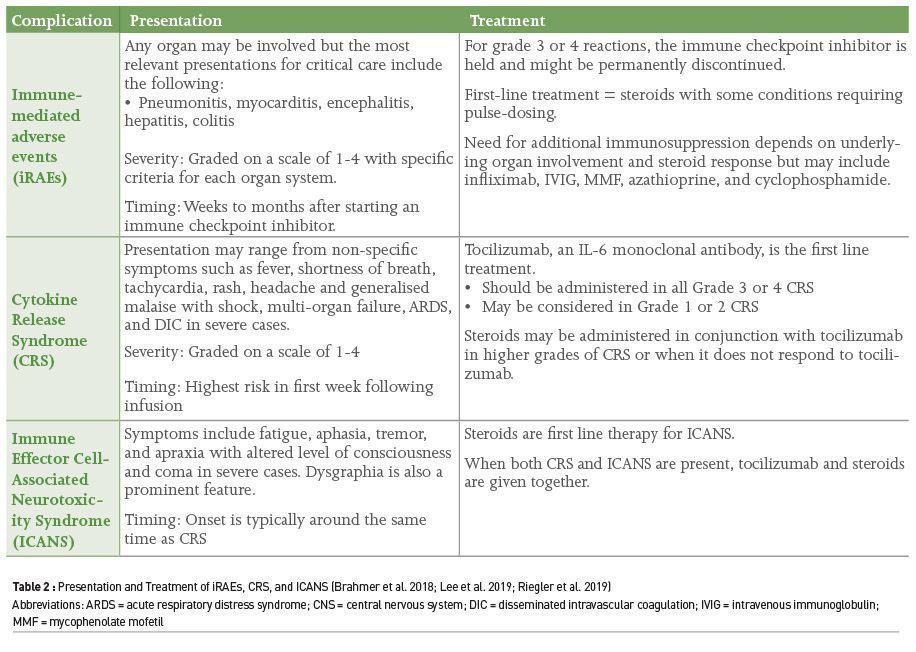
ICIs are primarily used in solid tumours but pembrolizumab, which is a monoclonal antibody against the programmed death-1 (PD-1) receptor, is approved for Hodgkin lymphoma and primary mediastinal large B-cell lymphoma (Twomey et al. 2021). ICIs allow the patient’s T-cells to target cancer by blocking checkpoints in the immune response. However, this upregulation of the immune system may result in side-effects known as immune-related adverse events (irAEs), which can affect any organ system and may lead to death in severe cases (Wang et al. 2018).
In CAR T-cell therapy, the patient’s T-cells are collected and genetically modified to recognise and target antigens on cancer cells. This therapy has been approved for use in a variety of haematologic malignancies including several types of lymphoma, ALL, and multiple myeloma. Leveraging the immune response in this way can lead to an inflammatory cascade known as cytokine release syndrome (CRS), which may lead to shock and multiorgan failure in severe presentations. Neurologic sequelae known as immune effector cell-associated neurotoxicity syndrome (ICANS) may also occur either in conjunction with or independently from CRS.
An overview of the presentation and treatment of iRAEs, CRS, and ICANS is outlined in Table 2. Severity grades of CRS and ICANS have been defined in the literature and many centres with CART programmes have established guidelines surrounding thresholds for initiation of pharmacologic treatments to blunt inflammation (eg. corticosteroids) and thresholds for ICU admission. Decisions surrounding treatment should be made following a multidisciplinary discussion with the oncology team. It is also important to have a low threshold to treat empirically for sepsis, and to rule out other mimickers of CRS such as haemophagocytic lymphohistiocytosis (HLH), and TLS.
Complications Following Allogeneic Haematopoietic Stem Cell Transplantation
Although outcomes have improved overall in recent decades for critically ill patients with haematologic malignancy, ICU mortality among patients who have undergone an allogeneic haematopoietic stem cell transplant (HSCT) remains high (Darmon et al. 2019). There are a range of complications that may result in critical illness and are important to have in the differential diagnosis with respiratory failure as the most common reason for ICU admission (Bayraktar et al. 2013; Kew et al. 2006). The post-transplant course is generally divided into the pre-engraftment stage (day 0-30), early post-engraftment stage (day 31-100) and late post-engraftment stage (after 100 days) with variable risks in each time frame. If a patient presents with critical illness after having undergone transplantation, understanding these three distinct timepoints is critical to understanding the mechanism of immunosuppression they are exposed to. This will inform which potential infectious organisms they are susceptible to as well as help develop the non-infectious differential. The major infectious considerations in each of these periods are outlined in Figure 2. Although patients may develop respiratory failure related to these infectious causes, there are a range of non-infectious aetiologies for respiratory failure in this population (Table 3). Close communication with the haemato-oncology team and oncologic- infectious disease services can help guide identifying cause and empiric management.

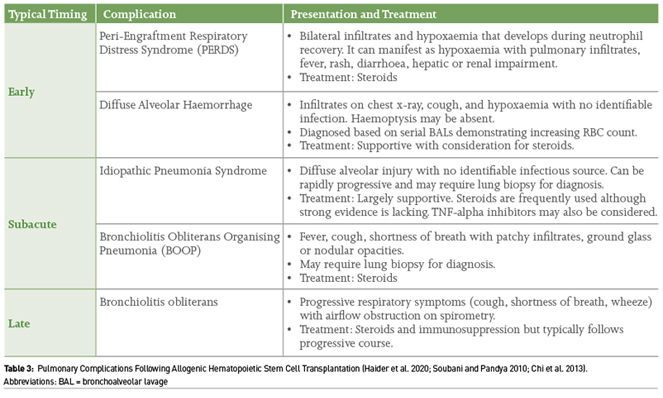
In addition to infectious and respiratory complications, other post-transplant complications to be aware of include acute graft versus host disease (GVHD), veno-occlusive disease (VOD), and neurologic complications. Acute GVHD traditionally occurs in the first 100 days post-transplant and most commonly manifests with skin involvement, gastrointestinal symptoms, and hyperbilirubinaemia although other organs may be involved. The mainstay of treatment is steroids with the potential addition of other immunosuppressive agents or the kinase inhibitor ruxolitinib in refractory disease (DiMaggio 2020). VOD, also called sinusoidal obstructive syndrome (SOS) is caused by obstruction of the hepatic venules and sinusoids in the liver. The aetiology is felt to be due to hepatic endothelial damage from transplant conditioning medications. VOD presents with abdominal pain, hepatomegaly, jaundice, ascites, weight gain and thrombocytopenia that typically presents in the pre-engraftment phase. Definitive diagnosis is often made by biopsy as imaging techniques are not sufficient. Treatment includes supportive care (paracentesis, monitoring fluid balance) with the addition of defibrotide in severe cases. For refractory disease which carries a significant mortality risk, steroids, transhepatic portosystemic shunt (TIPS), and liver transplantation may be considered and should involve the gastroenterology or hepatology team at an early stage (Senzolo et al. 2007). Finally, patients may also develop altered level of consciousness which can be related to infectious encephalities, drug toxicity (e.g., fludarabine neurotoxicity), intracranial haemorrhage, posterior reversible encephalopathy syndrome, neuro-GVHD, post-transplant lymphoproliferative disorder, or progressive multifocal leukencephalopathy, among others (Pruitt et al. 2013).
Conclusion
Patients with haematologic malignancies presenting with critical illness represent a unique population with specific syndromes. Early identification of aetiologies of critical illness and prompt initiation of appropriate critical care support is essential to their improved outcomes. Given their complexity, a multidisciplinary approach to their management with close collaboration between haemato-oncology and critical care is needed.
Conflict of Interest
None.
References:
Ali AM, Mirrakhimov AE, Abboud CN, Cashen AF (2016) Leukostasis in adult acute hyperleukocytic leukemia: a clinician's digest. Hematol Oncol, 34(2):69-78.
Bayraktar UD, Shpall EJ, Liu P et al. (2013) Hematopoietic cell transplantation-specific comorbidity index predicts inpatient mortality and survival in patients who received allogeneic transplantation admitted to the intensive care unit. J Clin Oncol, 31(33):4207-4214.
Bewersdorf JP, Zeidan AM (2020a) Hyperleukocytosis and Leukostasis in Acute Myeloid Leukemia: Can a Better Understanding of the Underlying Molecular Pathophysiology Lead to Novel Treatments? Cells, 9(10).
Bewersdorf JP, Giri S, Tallman MS et al. (2020b) Leukapheresis for the management of hyperleukocytosis in acute myeloid leukemia-A systematic review and meta-analysis. Transfusion, 60(10):2360-2369.
Brahmer JR, Lacchetti C, Thompson JA (2018) Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: American Society of Clinical Oncology Clinical Practice Guideline Summary. J Oncol Pract, 14(4):247-249.
Castillo JJ, Garcia-Sanz R, Hatjiharissi E et al. (2016) Recommendations for the diagnosis and initial evaluation of patients with WaldenstromMacroglobulinaemia: A Task Force from the 8th International Workshop on WaldenstromMacroglobulinaemia. Br J Haematol, 175(1):77-86.
Cairo MS, Bishop M (2004) Tumour lysis syndrome: new therapeutic strategies and classification. Br J Haematol, 127(1):3-11.
Chaudhary K, Gupta A, Wadhawan S (2012) Anesthetic management of superior vena cava syndrome due to anterior mediastinal mass. J Anaesthesiol Clin Pharmacol, 28(2):242-246.
Coombs CC, Tavakkoli M, Tallman MS (2015) Acute promyelocytic leukemia: where did we start, where are we now, and the future. Blood Cancer J, 5:e304.
Crawford J, Cox EB, Cohen HJ (1985) Evaluation of hyperviscosity in monoclonal gammopathies. Am J Med, 79(1):13-22.
Darmon M, Bourmaud A, Georges Q et al. (2019) Changes in critically ill cancer patients' short-term outcome over the last decades: results of systematic review with meta-analysis on individual data. Intensive Care Med, 45(7):977-987.
DiMaggio E (2020) Acute Graft-versus-Host Disease: Emerging Insights and Updates into Detection, Prevention, and Treatment. Pharmacotherapy, 40(8):788-807.
Dimopoulos MA, Kastritis E (2019) How I treat Waldenstrom macroglobulinemia. Blood, 134(23):2022-2035.
Duceau B, Picard M, Pirracchio R et al. (2019) Neutropenic Enterocolitis in Critically Ill Patients: Spectrum of the Disease and Risk of Invasive Fungal Disease. Crit Care Med, 47(5):668-676.
Durani U, Shah ND, Go RS (2017) In-Hospital Outcomes of Tumor Lysis Syndrome: A Population-Based Study Using the National Inpatient Sample. Oncologist, 22(12):1506-1509.
Ferreyro BL, Scales DC, Wunsch H et al. (2021) Critical illness in patients with hematologic malignancy: a population-based cohort study. Intensive Care Med, 47(10):1104-1114.
Freifeld AG, Bow EJ, Sepkowitz KA et al. (2011) Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the infectious diseases society of america. Clin Infect Dis, 52(4):e56-93.
Friedman T, Quencer KB, Kishore SA et al. (2017) Malignant Venous Obstruction: Superior Vena Cava Syndrome and Beyond. Semin InterventRadiol, 34(4):398-408.
Gorschluter M, Mey U, Strehl J et al. (2005) Neutropenic enterocolitis in adults: systematic analysis of evidence quality. Eur J Haematol, 75(1):1-13.
Haider S, Durairajan N, Soubani AO (2020) Noninfectious pulmonary complications of haematopoietic stem cell transplantation. Eur Respir Rev, 29(156).
Hiemenz JW (2009) Management of infections complicating allogeneic hematopoietic stem cell transplantation. Semin Hematol, 46(3):289-312.
Jones GL, Will A, Jackson GH et al. (2015) British Committee for Standards in H. Guidelines for the management of tumour lysis syndrome in adults and children with haematological malignancies on behalf of the British Committee for Standards in Haematology. Br J Haematol, 169(5):661-671.
Klastersky J, de Naurois J, Rolston K et al. (2016) Management of febrile neutropaenia: ESMO Clinical Practice Guidelines. Ann Oncol, 27(suppl 5):v111-v118.
Kew AK, Couban S, Patrick W et al. (2006) Thompson K, White D. Outcome of hematopoietic stem cell transplant recipients admitted to the intensive care unit. Biol Blood Marrow Transplant, 12(3):301-305.
Kochanek M, Schalk E, von Bergwelt-Baildon M et al. (2019) Management of sepsis in neutropenic cancer patients: 2018 guidelines from the Infectious Diseases Working Party (AGIHO) and Intensive Care Working Party (iCHOP) of the German Society of Hematology and Medical Oncology (DGHO). Ann Hematol, 98(5):1051-1069.
Kuderer NM, Dale DC, Crawford J et al. (2006) Mortality, morbidity, and cost associated with febrile neutropenia in adult cancer patients. Cancer, 106(10):2258-2266.
Lanciego C, Pangua C, Chacon JI et al. (2009) Endovascular stenting as the first step in the overall management of malignant superior vena cava syndrome. AJR Am J Roentgenol, 193(2):549-558.
Lee DW, Santomasso BD, Locke FL et al. (2019) ASTCT Consensus Grading for Cytokine Release Syndrome and Neurologic Toxicity Associated with Immune Effector Cells. Biol Blood Marrow Transplant, 25(4):625-638.
Lehmann S, Deneberg S, Antunovic P et al. (2017) Early death rates remain high in high-risk APL: update from the Swedish Acute Leukemia Registry 1997-2013. Leukemia, 31(6):1457-1459.
Leow L, Sampath HK, Yong KJ et al. (2021) Rescue extracorporeal membrane oxygenation for massive anterior mediastinal masses. J Artif Organs.
Levi M, Toh CH, Thachil J, Watson HG (2009) Guidelines for the diagnosis and management of disseminated intravascular coagulation. British Committee for Standards in Haematology. Br J Haematol, 145(1):24-33.
Mamez AC, Raffoux E, Chevret S et al. (2016) Pre-treatment with oral hydroxyurea prior to intensive chemotherapy improves early survival of patients with high hyperleukocytosis in acute myeloid leukemia. Leuk Lymphoma, 57(10):2281-2288.
Marbello L, Ricci F, Nosari AM et al. (2008) Outcome of hyperleukocytic adult acute myeloid leukaemia: a single-center retrospective study and review of literature. Leuk Res, 32(8):1221-1227.
Matuszkiewicz-Rowinska J, Malyszko J (2020) Prevention and Treatment of Tumor Lysis Syndrome in the Era of Onco-Nephrology Progress. Kidney Blood Press Res, 45(5):645-660.
Mehta J, Singhal S (2003) Hyperviscosity syndrome in plasma cell dyscrasias. Semin ThrombHemost, 29(5):467-471.
Mhaskar R, Clark OA, Lyman G et al. (2014) Colony-stimulating factors for chemotherapy-induced febrile neutropenia. Cochrane Database Syst Rev, (10):CD003039.
Micol JB, Raffoux E, Boissel N et al. (2014) Management and treatment results in patients with acute promyelocytic leukaemia (APL) not enrolled in clinical trials. Eur J Cancer, 50(6):1159-1168.
Mignard X, Biard L, Lemiale V et al. (2019) Granulocyte colony-stimulating factor and respiratory status of critically ill neutropenic patients with hematologic malignancies. Leuk Lymphoma, 60(5):1156-1163.
Montesinos P, Bergua JM, Vellenga E et al. (2009) Differentiation syndrome in patients with acute promyelocytic leukemia treated with all-trans retinoic acid and anthracycline chemotherapy: characteristics, outcome, and prognostic factors. Blood, 113(4):775-783.
Patel KS, Lau JE, Zembillas AS, Gallagher EM (2017) Single 4.5 mg fixed-dose of rasburicase for hyperuricemia associated with tumor lysis syndrome. J Oncol Pharm Pract, 23(5):333-337.
Porcu P, Cripe LD, Ng EW et al. (2000) Hyperleukocytic leukemias and leukostasis: a review of pathophysiology, clinical presentation and management. Leuk Lymphoma, 39(1-2):1-18.
Pruitt AA, Graus F, Rosenfeld MR (2013) Neurological complications of transplantation: part I: hematopoietic cell transplantation. Neurohospitalist, 3(1):24-38.
Rachapalli V, Boucher LM (2014) Superior vena cava syndrome: role of the interventionalist. Can Assoc Radiol J, 65(2):168-176.
Riegler LL, Jones GP, Lee DW (2019) Current approaches in the grading and management of cytokine release syndrome after chimeric antigen receptor T-cell therapy. Ther Clin Risk Manag, 15:323-335.
Rodrigues FG, Dasilva G, Wexner SD (2017) Neutropenic enterocolitis. World J Gastroenterol, 23(1):42-47.
Rollig C, Ehninger G (2015) How I treat hyperleukocytosis in acute myeloid leukemia. Blood, 125(21):3246-3252.
Sahin U, Toprak SK, Atilla PA et al. (2016) An overview of infectious complications after allogeneic hematopoietic stem cell transplantation. J Infect Chemother, 22(8):505-514.
Saillard C, Zafrani L, Darmon M et al. (2018) The prognostic impact of abdominal surgery in cancer patients with neutropenic enterocolitis: a systematic review and meta-analysis, on behalf the Groupe de Recherche en Reanimation Respiratoire du patient d'Onco-Hematologie (GRRR-OH). Ann Intensive Care, 8(1):47.
Sanz MA, Fenaux P, Tallman MS et al. (2019) Management of acute promyelocytic leukemia: updated recommendations from an expert panel of the European LeukemiaNet. Blood, 133(15):1630-1643.
Senzolo M, Germani G, Cholongitas E et al. (2007) Veno occlusive disease: update on clinical management. World J Gastroenterol, 13(29):3918-3924.
Shallis RM, Stahl M, Wei W et al. (2020) Patterns of care and clinical outcomes of patients with newly diagnosed acute myeloid leukemia presenting with hyperleukocytosis who do not receive intensive chemotherapy. Leuk Lymphoma, 61(5):1220-1225.
Soubani AO, Pandya CM (2010) The spectrum of noninfectious pulmonary complications following hematopoietic stem cell transplantation. Hematol Oncol Stem Cell Ther, 3(3):143-157.
Stahl M, Shallis RM, Wei W et al. (2020) Management of hyperleukocytosis and impact of leukapheresis among patients with acute myeloid leukemia (AML) on short- and long-term clinical outcomes: a large, retrospective, multicenter, international study. Leukemia, 34(12):3149-3160.
Stefanski M, Jamis-Dow C, Bayerl M et al. (2016) Chest radiographic and CT findings in hyperleukocytic acute myeloid leukemia: A retrospective cohort study of 73 patients. Medicine (Baltimore), 95(44):e5285.
Stone MJ, Bogen SA (2012) Evidence-based focused review of management of hyperviscosity syndrome. Blood, 119(10):2205-2208.
Tallman MS, Altman JK (2009) How I treat acute promyelocytic leukemia. Blood, 114(25):5126-5135.
Taplitz RA, Kennedy EB, Bow EJ et al. (2018) Outpatient Management of Fever and Neutropenia in Adults Treated for Malignancy: American Society of Clinical Oncology and Infectious Diseases Society of America Clinical Practice Guideline Update. J Clin Oncol, 36(14):1443-1453.
Tomblyn M, Chiller T, Einsele H et al. (2009) Guidelines for preventing infectious complications among hematopoietic cell transplantation recipients: a global perspective. Biol Blood Marrow Transplant, 15(10):1143-1238.
Truong TH, Beyene J, Hitzler J et al. (2007) Features at presentation predict children with acute lymphoblastic leukemia at low risk for tumor lysis syndrome. Cancer, 110(8):1832-1839.
Twomey JD, Zhang B (2021) Cancer Immunotherapy Update: FDA-Approved Checkpoint Inhibitors and Companion Diagnostics. AAPS J, 23(2):39.
Wang DY, Salem JE, Cohen JV et al. (2018) Fatal Toxic Effects Associated With Immune Checkpoint Inhibitors: A Systematic Review and Meta-analysis. JAMA Oncol, 4(12):1721-1728.
Weaver A, Rubinstein S, Cornell RF (2020) Hyperviscosity Syndrome in Paraprotein Secreting Conditions Including Waldenstrom Macroglobulinemia. Front Oncol, 10:815.
Yilmaz M, Kantarjian H, Ravandi F (2021) Acute promyelocytic leukemia current treatment algorithms. Blood Cancer J, 11(6):123.





















