Sepsis is a life-threatening condition in children. The lack of a clear definition leads to a heterogeneous diagnosis that prevents obtaining fully reliable epidemiological data. In the study conducted by Weiss et al. (2015) the Paediatric Intensive Care Unit (PICU) prevalence worldwide was 8.2% (Europe 6.2%), and mortality was 25%, and did not differ by age or between developed and resource-limited countries. For community-acquired sepsis in European PICUs, mortality was 6%, increasing to 10% in the presence of septic shock. Of the survivors, 31% were discharged with disability (Boeddha et al. 2018).
In 2005, the Barcelona Consensus Conference constituted by paediatric experts published the paediatric definitions, which were based on systemic inflammatory response syndrome (SIRS) criteria (Goldstein et al. 2005), approaching the adult definitions of 1991 and 2001. To date, these criteria have been the most widely used to define and classify septic patients in children.
Use of the SIRS criteria has been criticised in recent years because of their non-specificity. On the other hand, there are patients who may not have two or more SIRS criteria and suffer an infection with organ failure (Weiss et al. 2015; Brown et al 2015).
The third international consensus definitions for sepsis for adults (Sepsis-3) (Singer et al. 2016) agreed on new criteria based on organ dysfunction. Sepsis is now defined as the organ dysfunction that appears due to the inflammatory host response to an infection (10% death risk). The Sepsis-3 definitions aim to simplify the diagnosis and allow premature detection of patients with organ failure, at a higher risk of death. Organ dysfunction is measured as an acute increase of two points in the Sequential Organ Failure Assessment Scale (SOFA) due to an infection. Blood tests are necessary to define SOFA, so it can be a non-optimal tool outside the ICU. For this reason the use of quick SOFA (qSOFA) has been proposed.
As far as the paediatric patient is concerned, what influence can the Sepsis-3 definitions have? A consensus has not been published since 2005, and the latest paediatric Surviving Sepsis Campaign (SSC) guidelines have not yet been published; the last update was published in 2013. Presumably, the inclusion of the concept of organ dysfunction in the definition of sepsis will be shared by the community of paediatricians. Efforts are focused on finding a definition that involves a premature diagnosis with prognostic implications based on the organ dysfunction as well as in the adult patient. The SOFA score has not been designed for use in children, a population that has different vital signs according to age. At present, attempts are being made to adapt the definitions established for adults through a paediatric score.
The use of Sepsis-3 definitions in children was feasible and showed promising results. Schlapbach and colleagues (2017) demonstrated that the two SIRS variables-based sepsis criteria had poor specificity to discriminate children with infection at substantially higher mortality risk. Moreover, age-adapted SOFA and Paediatric Logistic Organ Dysfunction-2 score (PELOD-2) had significantly greater prognostic accuracy for in-hospital mortality. Their findings indicate that age-specific translation of Sepsis-3 definitions to critically ill children using validated measures of organ dysfunction should be considered in the next revision of paediatric sepsis definitions. In contrast, the performance of qSOFA to identify patients with organ dysfunction at risk for worse outcomes was poor, and may not be of sufficient clinical value to be recommended as a screening tool for paediatric age groups within the ICU. Leclerc and colleagues (2017) concluded that in children admitted to PICU with suspected infection, PELOD-2 score on day 1 was highly predictive of PICU mortality, suggesting its use to standardise definitions and diagnostic criteria of paediatric sepsis. The SOFA score was adapted and validated with age-adjusted cutoffs in critically ill children: paediatric SOFA (pSOFA). Sepsis-3 definitions were assessed in children with confirmed or suspected infection (Matics et al. 2017). All these scores might be more useful for prognosis than for diagnosis of sepsis.
Regarding biomarkers, values of C-reactive protein and procalcitonin have been included in the usual management although their sensitivity and specificity is lower than desired. Lactate continues to be valid because its elevation is related to organ dysfunction and worse prognosis; monitoring its levels can also assess the therapeutic response. New hopes are focused on other biological markers, such as adrenomoduline (Jordan et al. 2014) or pro-inflammatory and anti-inflammatory cytokines (Polic et al. 2017), to help us in diagnosis, prognosis, and follow-up. We do not have a perfect biomarker to date; perhaps in the future a panel of different biomarkers may be the key (Lamping et al. 2018).
Treatment
The latest SSC guidelines that include paediatric management date from 2012 (Dellinger et al. 2013). The American College of Critical Care Medicine (ACCM) published in 2017 the guide for haemodynamic support in neonates and children with septic shock (Davis et al. 2017).
Despite the dissemination of the previous guidelines, some studies demonstrated incomplete adherence to recommendations (Moresco et al. 2018). Consequently quality improvement studies were designed, in order to trigger rapid clinician evaluation and implementation of appropriate resuscitation efforts (Esteban et al. 2017; Cruz et al 2011). The new ACCM guidelines recommend that each institution implements its own adopted or home-grown bundles resuscitation and stabilisation bundle to drive adherence to consensus best practice.
The first hour of resuscitation
The goals of the first hour should be to maintain the airway, oxygenation, and ventilation; Maintain or restore circulation, capillary refill, normal pulses, urine output ≥ 1 mL/kg/hr, normal mental status, normal blood pressure for age; and restore appropriate heart rate (HR).
During initial resuscitation the achievement of objectives is evaluated by minimal invasive monitoring. Echocardiography is considered an appropriate noninvasive tool to evaluate myocardial contractility and intravascular volume, to direct resuscitation goals and therapeutic endpoints.
Supplemental high-flow oxygen should be provided. Children with persistent or worsening shock should be intubated and receive mechanical ventilation to eliminate breathing effort and improve oxygenation and organ perfusion. In most cases, there is time for fluid resuscitation and starting a peripheral inotropic infusion before airway management is needed. Patients with these characteristics are vulnerable to the haemodynamic effects of sedatives (Li et al. 2016), emphasising the importance of initial resuscitation prior to airway instrumentation. The use of ketamine with atropine is considered to be the induction regimen which best promotes cardiovascular integrity. The use of etomidate is discouraged given its effects on adrenal function. Other options to consider are fentanyl and remifentanil or benzodiazepines titrated with small doses. Barbiturates, inhalational agents or propofol are not appropriate. Neuromuscular blocking agents may facilitate intubation.
Vascular access should be rapidly accomplished. Portable near-infrared imaging devices may assist in peripheral vascular access. Establish intraosseous access if peripheral intravenous line access cannot be attained in 5-10 minutes. Establishing a central venous catheter during the initial resuscitation should not delay or compromise the resuscitation efforts. Ultrasound guidance may facilitate placement of central catheters.
Extract sample for blood analysis and culture when a vascular access is accomplished. Initiate antibiotic therapy as soon as possible after obtaining cultures and always during the first hour of clinical suspicion.
Fluid resuscitation should start immediately unless hepatomegaly, rales, or a cardiac gallop are present. If these signs are present, the patient may not require fluid boluses and instead, inotropic support is recommended. The harm of fluid boluses at the initial haemodynamic resuscitation is a concern (Frazier et al. 2015). Fluid infusion (crystalloids or colloids) is best initiated with boluses of 20 mL/kg, titrated to assuring signs of restored circulation and normal HR as commented above. Initial (first hour) volume resuscitation requirements commonly are 40–60 mL/kg. Specific evaluation after each bolus for signs of fluid overload and simple algorithms are needed to support healthcare providers to choose which patients could be harmed and which will benefit from fluid boluses (Ford et al. 2012; Parker et al. 2016).
Patients who do not respond rapidly to initial fluid boluses should be considered for invasive haemodynamic monitoring. In the fluid refractory patient, begin a peripheral epinephrine, while establishing a central venous catheter. Dopamine, epinephrine, or norepinephrine can be administered as a first-line drug as indicated by haemodynamic state when a central line is available.
Beyond the first hour (PICU haemodynamic support)
Monitoring should be intensified by adding invasive instruments to the clinical objectives: central venous access, arterial pressure monitoring and a modality to assess cardiac output (CO) are recommended.
Goal-directed therapy to achieve:
Perfusion pressure (mean arterial pressure (MAP)-central venous pressure (CVP) or MAP- intra-abdominal pressure (IAP)) appropriate for age. It is considered necessary for organ perfusion.
Venous oxygen saturation (ScvO2) greater than 70% is associated with improved outcome. ScvO2 saturation can be used as an indirect indicator of whether CO is adequate to meet tissue metabolic demand.
Cardiac index (CI) greater than 3.3 and less than 6.0 L/min/m2 may result in improved survival. Contrary to the adult experience, low CO, not low systemic vascular resistance (SVR), is associated with mortality in paediatric septic shock.
Normal INR, anion gap, and lactate are also objectives at this point.
Fluid losses and persistent hypovolaemia secondary to diffuse capillary leak can continue for days. Ongoing fluid replacement should be monitored carefully to evaluate bolus response. Crystalloid is the fluid of choice in patients with haemoglobin greater than 10 g/dL. Red blood cells transfusion can be given to children with haemoglobin less than 10 g/dL. Fresh frozen plasma is recommended for patients with prolonged INR (infusion, not bolus). Following shock resuscitation, diuretics/peritoneal dialysis/high flux continuous renal replacement therapy can be used to remove fluid in patients who are fluid overloaded and unable to maintain fluid balance.
Septic shock represents a dynamic process so haemodynamic drugs selected and their infusion dose may need to be changed over time. Frequent re-evaluation of haemodynamic parameters is recommended as haemodynamic support can be required for days.
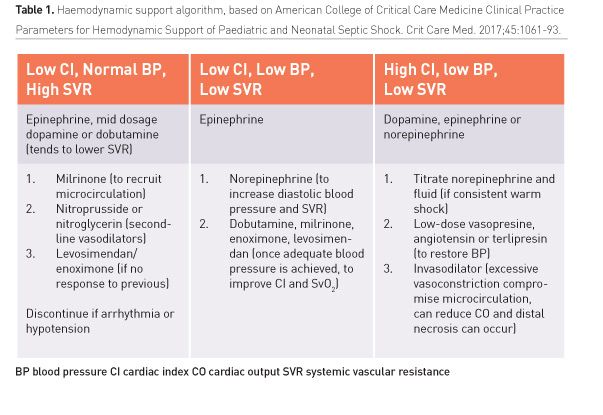
For children with “fluid-resistant shock” adrenaline is the initial inotrope (dopamine loses prominence), except in a clear pattern of low resistance and hypotension when noradrenaline may be the first drug. Catecholamine-resistant shock can present with low CO/high SVR, high CO/low SVR, or low CO/low SVR shock (Table 1), which will determine following treatment.
Angiotensin II has been recently reported as an effective treatment in patients with refractory vasodilatory shock (Khanna et al. 2017). Due to the lack of specific paediatric trials, its use in children remains prudent despite exceptional experiences (Yunge et al. 2000).
Consider hydrocortisone in refractory shock in children at risk of adrenal insufficiency. Extracorporeal membrane oxygenation (ECMO) is a viable therapy for refractory septic shock in neonates and children (Solé et al. 2018). Paediatric and adult patients with sepsis have lower survival (historically ≤ 50%) than neonates (80% + survival), but experienced ECMO centres are now reporting survival rates approaching 75% (MacLaren et al. 2011).
Outcome benefits of CRRT (management of fluid overload, acute kidney injury, clearance of lactate or inflammatory, etc.), either alone or in tandem with ECMO, should be considered in paediatric sepsis.
Regarding blood purification, in 2010 the American Society of Apheresis gave a category III recommendation, which is “Optimum role of apheresis therapy is not established. Decision-making should be individualized”. Therapeutic plasma exchange could be considered as a strategy to reverse multi-organ dysfunction syndrome especially in patients with significant coagulopathy.
Conclusion
New studies in children to diagnose and classify sepsis according to organ failure-based scores are promising, and probably a new consensus will approach adult definitions. Adrenaline and noradrenaline will play a major role in shock treatment in children, dopamine being less recommended. We expect the publication of the new paediatric SSC guidelines and new consensus in the following months.
Abbreviations
ACCM American College of Critical Care Medicine
BP blood pressure
CI cardiac index
CO cardiac output
CRRT continuous renal replacement therapy
CVP central venous pressure
ECMO extracorporeal membrane oxygenation
HR heart rate
IAP intra-abdominal pressure
ICU intensive care unit
INR international normalised ratio
MAP mean arterial pressure
PELOD-2 Paediatric Logistic Organ Dysfunction-2 score
PICU Paediatric Intensive Care Unit
pSOFA paediatric Sequential Organ Failure Assessment scale
qSOFA quick Sequential Organ Failure Assessment scale
RBC red blood cells
ScvO2 Venous oxygen saturation
SIRS Systemic Inflammatory Response Syndrome
SOFA Sequential Organ Failure Assessment scale
SSC Surviving Sepsis Campaign
SVR systemic vascular resistance