ICU Management & Practice, Volume 21 - Issue 6, 2021
This review focuses on different techniques to reverse direct oral anticoagulants, either with specific or non-specific agents.
Direct oral anticoagulants (DOACs) are now standing for Vitamin K antagonists in many clinical settings. They are recognised as a major step forward for our patients. Their efficacy has been confirmed in venous thromboembolism prophylaxis and treatment, in atrial fibrillation and even in acute coronary syndromes (Stensballe and Møller 2018). The biological monitoring is now widely available, and the interactions and contraindications are well known. The ratio of efficacy/safety is widely positive. Unfortunately, the global picture is still fuzzy as far as the reversion of these agents is concerned (Jaspers et al. 2021). Only two specific antidotes are approved, but the compound which is mostly used (prothrombin complex concentrate), is a non-specific reversal agent. This review will focus on different techniques to reverse DOACs.
There are several ways to control the anticoagulant activity of these agents while acting either on absorption, on the mechanism of action, or on elimination (Crowther and Crowther 2015).
Only activated charcoal deals with absorption. Wang et al. (2014) have given 50 grammes of activated charcoal to healthy volunteers who had been previously treated with apixaban either two hours or six hours before charcoal was given. Activated charcoal was able to decrease the apixaban elimination half-life from 13 hours down to five hours. The limit of this compound is that it has to be given in a rather short time slot i.e. six hours max.
Another way to decrease DOAC concentration is to act on elimination with haemodialysis. This technique has been shown to be effective in decreasing the plasma concentration of dabigatran by 50% after a four-hour procedure (Khadzhynov et al. 2013). Of note, after the interruption of the dialysis, a rebound of the dabigatran concentration has been observed. It has to be understood that this technique is only available for dabigatran which binding to the proteins is weak. Even if it is usually implemented in an intensive care environment, it may be difficult and dangerous to insert a very large catheter in an old patient overdosed with dabigatran.
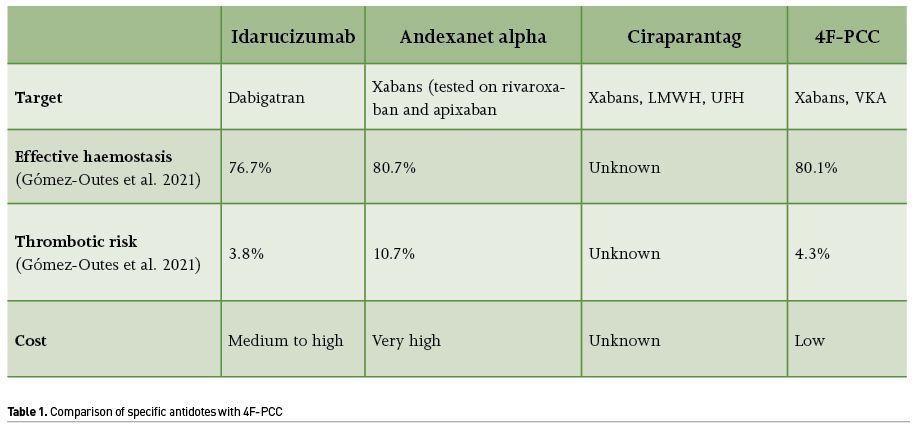
Prothrombin Complex Concentrates (PCC) and activated Prothrombin Complex Concentrates (FEIBA®) interfere with the mechanism of action. They have been tested with various doses and have shown conflicting results in different animal models (Godier et al. 2012) and healthy volunteers. They are now being used by clinicians in bleeding patients on a non-evidence basis. Several series, especially in neurology/neurosurgery patients have shown a better outcome in patients treated with four factor-PCC (4F-PCC as compared with nothing) (Grandhi et al. 2015). In the GIHP-NACO registry, 4F-PCC and aPCC have been shown to partially or totally control bleeding in patients treated with DOACs (Albaladejo et al. 2017). Majeed et al. (2017) and Schulman and colleagues (2018) have published two short series of bleeding patients treated with 4F-PCC with interesting results. Ten case series with 340 patients as a total have been reviewed by Piran et al. (2019). The pooled percentage of patients with effective management of major bleeding was 69% (95% confidence interval [CI], 61-76) in two studies using the International Society on Thrombosis and Haemostasis (ISTH) criteria and 77% (95% CI, 63-92) in eight studies that did not use the ISTH criteria; all-cause mortality was 16% (95% CI, 7-26), and thromboembolism rate was 4% (95% CI, 1-8). The obvious limit of such observational series is that it is difficult to determine whether 4F-PCC in addition to cessation of direct oral FXa inhibitor is more effective than cessation of direct oral FXa inhibitor alone in patients with direct FXa inhibitor–related major bleeding.
The same group has studied the efficacy/safety ratio of 4F-PCC (26 U/Kg) in 21 non-bleeding anti-Xa-treated patients who were about to undergo an emergent surgery or an invasive procedure (Piran et al. 2018). The efficacy of PCC was good in 18 patients, allowing a normal haemostasis with no thromboembolic events. Barzilai et al. (2019) have reported the results of a slightly larger study performed in two tertiary hospitals. Sixty-two apixaban or rivaroxaban patients were treated with 4F-PCC (26U/Kg) and assessed retrospectively. Bleeding during surgery was reported in 3 patients (5%), no patient required additional PCC, and 16 patients (26%) were transfused. The 30-day mortality and thrombosis rates were 21% and 3%, respectively.
Today, PCC is the most widely used agent for the reversal of xabans (anti-Xa agents). The median initial dose is close to 25 IU/Kg. A reinfusion might be necessary.
Activated PCC has also been used in some patients (Albaladejo et al. 2017). FEIBA® is a kind of haemostatic bomb which was initially dedicated to haemophiliacs with an inhibitor. It has been tested in vitro (Martin et al. 2015) and in small series of patients. Low doses (< 20 U/kg) to moderate (20–30 U/kg) doses appear to be effective, but with no follow-up for the side effects (Dager et al. 2018). Of note, this compound is very expensive.
Specific antidotes are also being developed. Three of them have already completed phase II and/or phase III studies:
- Idarucizumab (Praxbind®) is a fully humanised antibody fragment (Fab) which binds to the thrombin binding site of dabigatran hence inactivating the molecule. In healthy young and older volunteers, idarucizumab was associated with immediate, complete, and sustained reversal of dabigatran-induced anticoagulation (Glund et al. 2015). It was well tolerated with no unexpected or clinically relevant safety concerns. The global reversal duration lasted about 24 hours. The phase III study (REVERSE-AD) has included bleeding patients who have developed a serious bleeding event, or patients who require an urgent procedure (Pollack et al. 2017). The results including 503 patients show a complete reversal of the anticoagulant effect of dabigatran within minutes and 18% mortality (mainly unrelated to the antibody). The European (EMA) and U.S. (FDA) regulators have granted an approval for this compound, but further studies and a much larger number of patients are needed to be fully reassured. Nevertheless, this antibody may save lives. Some reinjections may be needed as a rebound in dabigatran concentration has been reported (Hegemann et al. 2018).
- Andexanet alpha is a recombinant modified human factor Xa protein that binds factor Xa inhibitors. This specific reversal agent is designed to neutralise the anticoagulant effects of both direct and indirect factor Xa inhibitors. Its half-life is short (less than 90 minutes) and the bolus has to be combined with a continuous IV infusion. Up to now, no data are available after a 6-hour administration. Andexanet has been shown to be effective in 101 healthy volunteers on a biological standpoint (Siegal et al. 2015). The ANNEXA-4 study has evaluated 352 patients who had acute major bleeding within 18 hours after administration of a factor Xa inhibitor (either apixaban or rivaroxaban) (Connolly et al. 2019). The patients received a bolus of andexanet, followed by a 2-hour infusion. Excellent or good haemostasis occurred in 204 of 249 patients (82%) who could be evaluated. Within 30 days, death occurred in 49 patients (14%) and a thrombotic event in 34 (10%). However, it has to be emphasised that the definition of an excellent or good haemostasis was debatable and that the very high thrombotic event rate was worrying. Furthermore, no data are available with a longer infusion period. Andexanet is effective, no doubt, but many undesirable side effects have led the FDA to issue a black box warning on thromboembolic risks, ischaemic risks, cardiac arrest, and sudden death: “Treatment with the agent has been associated with serious and life-threatening adverse events, including arterial and venous thromboembolic events, cardiac arrest, sudden deaths, and ischaemic events, such as myocardial infarction and ischemic stroke.” Is this what we expect from a specific reversal agent? Since, a study with a small number of patients has shown a lack of benefit of andexanet (Nederpelt et al. 2020) and a meta-analysis of studies on PCC, idarucizumab and andexanet has shown that the three agents developed the same efficacy for a comparable death toll but with a much higher thrombotic rate for andexanet (10.7% as compared to 4.3% for PCC and 3.8% for idarucizumab) (Gómez-Outes et al. 2021). A large study is ongoing, comparing andexanet to standard of care (mainly PCC). Results are awaited in 2023.
- Ciraparantag (PER977) is a small, synthetic, water-soluble, cationic molecule that is designed to bind specifically to unfractionated heparin, low-molecular-weight heparin, and to the new oral factor Xa inhibitors (xabans) (Chan and Weitz 2021). Ciraparantag directly binds to DOACs and to enoxaparin through non-covalent hydrogen bonds and charge–charge interactions Only phase 2 data are available for the moment. Ciraparantag provides a dose-related reversal of anticoagulation induced by steady-state dosing of apixaban or rivaroxaban. All doses of ciraparantag were well tolerated. The slow development of this compound probably relates to the almost impossibility to quantify its effect with conventional biological tests (aPTT, PT, antiXa level) because ciraparantag binds to the sodium citrate used for blood collection and to the reagents used to trigger clotting in these tests. In the reported studies, the anticoagulant effect of apixaban and rivaroxaban at steady state was assessed with the whole blood clot time (WBCT) (Ansell et al. 2021). Therefore, the quality of data is still weak and further studies with patients are needed.
- Other specific antidotes for xabans are being developed by several research groups and will be available in the near future, hopefully (Jourdi et al. 2018).
Conclusion
Conflict of Interest
None.
References:
Albaladejo P, Samama CM, Sié P et al. (2017) Management of Severe Bleeding in Patients Treated with Direct Oral Anticoagulants: An Observational Registry Analysis. Anesthesiology, 127:111–120.
Ansell J, Bakhru S, Laulicht BE et al. (2021) Ciraparantag reverses the anticoagulant activity of apixaban and rivaroxaban in healthy elderly subjects. European Heart Journal (published ahead of print).
Barzilai M, Kirgner I, Steimatzky A et al. (2020) Prothrombin Complex Concentrate before Urgent Surgery in Patients Treated with Rivaroxaban and Apixaban. Acta Haematologica, 143:266–271.
Chan NC, Weitz JI (2021) Ciraparantag as a potential universal anticoagulant reversal agent. European Heart Journal (published ahead of print).
Connolly SJ, Crowther M, Eikelboom JW et al. (2019) Full Study Report of Andexanet Alfa for Bleeding Associated with Factor Xa Inhibitors. The New England Journal of Medicine, 380:1326–1335.
Crowther M, Crowther MA (2015) Antidotes for novel oral anticoagulants: current status and future potential. Arteriosclerosis, Thrombosis, and Vascular Biology, 35:1736–1745.
Dager WE, Roberts A.J, Nishijima DK (2018) Effect of low and moderate dose FEIBA to reverse major bleeding in patients on direct oral anticoagulants. Thrombosis Research, 173:71–76.
Glund S, Stangier J, Schmohl M et al. (2015) Safety, tolerability, and efficacy of idarucizumab for the reversal of the anticoagulant effect of dabigatran in healthy male volunteers: a randomised, placebo-controlled, double-blind phase 1 trial. Lancet, 386:680–690.
Godier A, Miclot A, Bonniec BL et al. (2012) Evaluation of Prothrombin Complex Concentrate and Recombinant Activated Factor VII to Reverse Rivaroxaban in a Rabbit Model. Anesthesiology, 116: 94–102.
Gómez-Outes A, Alcubilla P, Calvo-Rojas G et al. (2021) Meta-Analysis of Reversal Agents for Severe Bleeding Associated With Direct Oral Anticoagulants. Journal of the American College of Cardiology, 77:2987–3001.
Grandhi R, Newman WC, Zhang X et al. (2015) Administration of 4-Factor Prothrombin Complex Concentrate as an Antidote for Intracranial Bleeding in Patients Taking Direct Factor Xa Inhibitors. World neurosurgery, 84:1956–1961.
Hegemann I, Ganter C, Widmer CC et al. (2018) Ongoing redistribution of dabigatran necessitates repetitive application of idarucizumab. British Journal of Anaesthesia, 121:505-508.
Jaspers T, Shudofsky K, Huisman MV et al. (2021) A meta‐analysis of andexanet alfa and prothrombin complex concentrate in the treatment of factor Xa inhibitor–related major bleeding. Research and Practice in Thrombosis and Haemostasis, 5:e12518.
Jourdi G, Gouin-Thibault I, Siguret V et al. (2018) FXa-α2-Macroglobulin Complex Neutralizes Direct Oral Anticoagulants Targeting FXa In Vitro and In Vivo. Thrombosis and Haemostasis, 118:1535–1544.
Khadzhynov D, Wagner F, Formella S et al. (2013) Effective elimination of dabigatran by haemodialysis. A phase I single-centre study in patients with end-stage renal disease. Thrombosis and Haemostasis, 109:596–605.
Majeed A, Ågren A, Holmström M et al. (2017) Management of rivaroxaban- or apixaban-associated major bleeding with prothrombin complex concentrates: a cohort study. Blood, 130:1706–1712.
Martin AC, Gouin-Thibault I, Siguret V et al. (2015) Multimodal assessment of non-specific hemostatic agents for apixaban reversal. Journal of thrombosis and haemostasis: JTH, 13:426–436.
Nederpelt C.J, Naar L, Sylvester KW et al. (2020) Evaluation of oral factor Xa inhibitor-associated extracranial bleeding reversal with andexanet alfa. Journal of Thrombosis and Haemostasis, 18:2532–2541.
Piran S, Gabriel C, Schulman S (2018) Prothrombin complex concentrate for reversal of direct factor Xa inhibitors prior to emergency surgery or invasive procedure: a retrospective study. Journal of Thrombosis and Thrombolysis, 45:486–495.
Piran S, Khatib R, Schulman S et al. (2019) Management of direct factor Xa inhibitor-related major bleeding with prothrombin complex concentrate: a meta-analysis. Blood advances, 3:158–167
Pollack CV, Reilly PA, Ryn JV et al. (2017) Idarucizumab for Dabigatran Reversal - Full Cohort Analysis. New England Journal of Medicine, 377:431–441.
Schulman S, Gross PL, Ritchie B et al. (2018) Prothrombin Complex Concentrate for Major Bleeding on Factor Xa Inhibitors: A Prospective Cohort Study. Thrombosis and Haemostasis, 118:842–851.
Siegal DM, Curnutte JT, Connolly SJ et al. (2015) Andexanet Alfa for the Reversal of Factor Xa Inhibitor Activity. New England Journal of Medicine, 373:2413–2424.
Stensballe J, Møller MH (2018) Ten things ICU specialists need to know about direct oral anticoagulants (DOACs). Intens Care Med, 370:847–4.
Wang X, Mondal S, Wang J et al. (2014) Effect of activated charcoal on apixaban pharmacokinetics in healthy subjects. American journal of cardiovascular drugs : drugs, devices, and other interventions, 14:147–154.





















