Introduction
Respiratory physiotherapy [RP] is an integral part of the multidisciplinary approach to patients hospitalised in intensive care units [ICUs] (Gosselink et al. 2008).
The survival rate of patients with life-threatening conditions who are admitted to an ICU has significantly increased through improvements in medical care. Unfortunately, for those who do survive, nearly half present long-term impairments in physical, cognitive and/or mental health, often due to post-intensive care syndrome [PICS] (Needham et al. 2012) and weakness acquired post-ICU, regardless of the diagnosis of admission to the ICU (Ohtake et al. 2018; Kamdar et al. 2017; Dinglas et al. 2017). Critical illness is frequently associated with long-term bed rest and inactivity, and it may lead to ICU-acquired muscle weakness [ICUAW] (Stevens et al. 2007), and other deleterious effects (Cirio et al. 2003), which in turn are strongly associated with increased morbidity, physical impairments and mortality, both at short and long-term (Herridge et al. 2011; Hermans et al. 2014; Unroe et al. 2010; Pandharipande et al. 2013). It has been recognised that physical and psychological recovery after a period of critical illness is slow and often incomplete (Wilson et al. 2018) and in certain conditions, it leads to a cognitive impairment comparable with Alzheimer's disease, especially in older patients (Pandharipande et al. 2013), and this decreases quality of life, for up to 5 years after their original illness (Heyland et al. 2005; Herridge et al. 2013; Iwashyna et al. 2010; Herridge et al. 2016a; Fan et al. 2014; Cox et al. 2009; Herridge et al. 2016b; Barnato et al. 2011; Turnbull et al. 2016; Chan et al. 2016; Kamdar et al. 2017).
Limiting the period of immobility and promoting early physiotherapy enhance recovery and influence or even prevent physical impairments and poor outcomes (Fuke et al. 2018; Schweickert et al. 2009; Schaller et al. 2016; Bourdin et al. 2010; Kayambu et al. 2013; Stiller et al. 2013). The aims of early physiotherapy consist in improving patient’s quality of life and preventing ICU-associated complications like deconditioning, ventilator dependency, and respiratory conditions. In addition, early rehabilitation can be extended beyond physical therapy to include cognitive therapy (Brummel et al. 2014).
Since respiratory pathologies are among the most common causes of admission to critical care, RP represents a fundamental part of the standard practice in ICU (Vincent et al. 2002; Berney et al. 2012). Respiratory physiotherapists provide various types of care, from acute respiratory interventions to rehabilitation. They treat both intubated and spontaneously breathing patients, and their key roles include management of respiratory, but also neurological and musculoskeletal complications (Pathmanathan et al. 2015).
The European Respiratory Society and European Society of Intensive Care Medicine Task Force on Physiotherapy for Critically Ill Patients in 2008 identified targets for physiotherapy for intensive care patients and summarised the literature regarding the available effective physiotherapy interventions (Gosselink et al. 2008). The Society of Critical Care Medicine recommends a comprehensive treatment for ICU survivors during all phases of recovery (Elliott et al. 2014; Hanekom et al. 2011). The authors developed a clinical management algorithm for early physical activity and mobilisation of intensive care patients in order to decrease clinical variability and to improve patient safety. Recently Sommers et al. provided a statement that explains safety criteria for the early mobilisation of intensive care patients (Sommers et al. 2015).
The purpose of this review is to update the evidence base regarding the application of RP to adult critically ill patients, with the goal of supporting and implementing clinical daily practice. This review provides a practical and feasible description of the main physiotherapeutic tools and strategies that can be applied to critically ill patients.
Method of systematic literature search
We performed a systematic computerised literature search of the electronic databases PubMed, Cochrane and Scopus for papers published in English in the last 20 years combining the following keywords and medical subject headings: intensive care, pulmonary rehabilitation, respiratory physiotherapy, critical illness, acquired weakness, early physiotherapy, and chest physiotherapy.No limitations in the search strategy were inserted, except for the exclusion of case reports, comments or letters.
Selection of patients to treat, monitoring and safety
Although several studies described the safety of early physiotherapy (Brummel et al. 2014; Schweickert et al. 2009; Morris et al. 2016), the application of techniques to critically ill patients is often difficult because of the critical respiratory conditions, necessitating medication and invasive equipment. Oftentimes these severe respiratory diseases are associated with haemodynamic compromise, and this limits the possibilities for applying the physiotherapy interventions even more. In addition, the medical situation of these critically ill patients can rapidly change. Therefore monitoring patients’ clinical status before and during physiotherapy sessions is of pivotal importance, first of all for patients’ safety. Changes in vital parameters (blood pressure, heart rate, saturation of peripheral oxygen, respiratory rate, dyspnoea) should be monitored during each treatment session, and in case of deterioration, the intervention should be stopped. However, adverse events are generally rare during RP (Zeppos et al. 2007; Bailey et al. 2007), with the most common ones represented by decreases in oxygen saturation, changes in mean arterial pressure (both increase and decrease), and arrhythmias. Risk factors for these adverse events are haemodynamic instability, presence of cardiac comorbidities and abnormalities of vital signs before intervention.
In order to increase the safety of RP, as part of the clinical reasoning process, every patient should be screened for the presence of absolute and relative contraindications, considering potential risks and benefits of each treatment. During this decision process, collaboration between all the members of the intensive care multidisciplinary team (intensivist, physiotherapist, nurse) is essential. The main contraindications to RP are shown in Table 1.
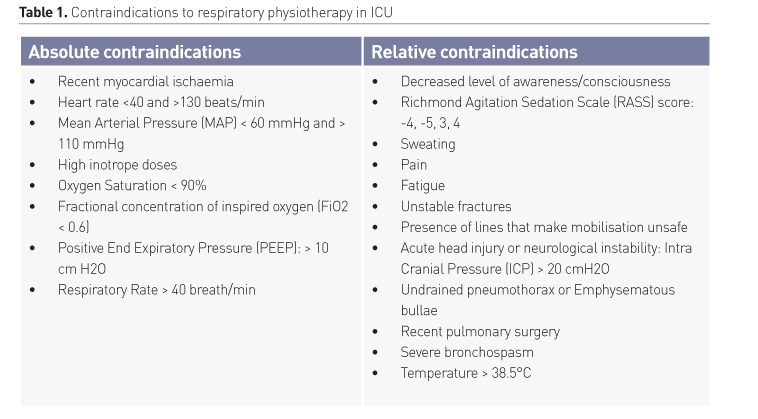
For every patient, we recommend assessment of global mental functions, responsiveness and consciousness and ability to cooperate. It should be noted that a significant reduction in vigilance, up to coma, does not represent an absolute contraindication to RP, because even in patients with low RASS score it’s possible to apply passive techniques. Other aspects to be evaluated are range of joint motion, muscle strength, and tone, sensory function, functional ability [e.g. transfers in and out of the bed, standing balance and walking] (Parry et al. 2017). In conscious patients, it’s important to monitor exertion during exercise (six-minute walk test, Borg score). All these parameters can be used to evaluate the impairments of every single patient and to programme tailored interventions.
Probably the biggest problems that clinicians have to face during RP are represented by barriers like staff and patient’s safety concerns, perceived workload, general lack of confidence that rehabilitation may improve outcomes, and lastly lack of updated guidelines.
Respiratory physiotherapy interventions
The RP interventions that are recommended for critically ill patients hospitalised in the ICU are presented in Table 2. For clinical practice, we divided the recommended physiotherapy interventions into two groups; interventions for patients who are able (active interventions) and interventions for those who are not able (passive interventions) to follow instructions, determined primarily by the level of consciousness.
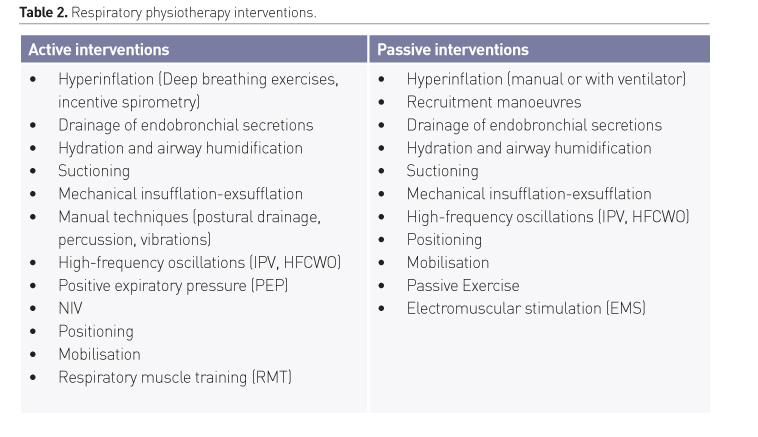
The main goal of RP is minimising the adverse effects of critical illness and intubation on the respiratory system (Goligher et al. 2019), restoring respiratory and physical independence, preventing the need for subsequent dependence on mechanical ventilation and subsequent hospitalisations and improving patient’s quality of life. First of all this is achieved by the reduction of patient’s dependency on the ventilator, reduction of secretion retention, atelectasis and pneumonia, maintenance or recruitment of lung volume, improvement of regional or global ventilation and compliance, improvement of ventilation/perfusion match, reduction of airway resistance and work of breathing, and optimisation of oxygenation. Not of less importance are the preservation and improvement of respiratory and peripheral muscle strength. Finally, an ICU early rehabilitation programme can generate net financial savings for the hospital, as shown by the financial model established by Lord et al. on actual experience and published data (Lord et al. 2013).
In the following section, we will discuss briefly the main interventions, dividing them into four major categories: techniques for lung hyperinflation, drainage of endobronchial secretions, mobilisation and muscular training.
1. Lung hyperinflation
Patients admitted to ICUs and affected by a respiratory disease frequently incur alterations of lung volumes, atelectasis, and reduction of respiratory flows that leads to increased pulmonary shunt, and impairment of gas exchange. All these detrimental effects are more pronounced in the case of mechanically ventilated patients.
Lung hyperinflation has been recognised as an important aspect to prevent atelectasis and to achieve recruitment of areas of pulmonary collapse leading to improved lung compliance and gas exchange (Stiller 2000). Moreover, it increases the movement of pulmonary secretions toward the central airways (Denehy 1999). This chest physiotherapy technique can be applied manually or through a ventilator.
Manual hyperinflation (MHI) involves the delivery via a manual resuscitator bag of transient tidal volumes larger than baseline (Hodgson et al. 1999); the delivery of a slow inspiration is usually followed by a 2–3 seconds inspiratory hold, and a fast, uninterrupted expiratory flow. MHI is commonly applied in patients under mechanical ventilation (MV), but this involves disconnecting the patient from the ventilator. It may stimulate cough and move the airway secretions toward the larger airways, from where they can be easily suctioned. MHI can prevent airway plugging and pulmonary collapse, and improve oxygenation and lung compliance (Paulus et al. 2011). There are different MHI bags available; some of them have PEEP valves within the circuit to ensure patients maintain PEEP and reduce de-recruitment and atelectotrauma. This is particularly useful in patients who are PEEP-dependent, such as patients with acute respiratory distress syndrome (ARDS). MHI can be performed using 100% oxygen, air alone, or a mixture of both. It is widely recognised that patients anaesthetised and ventilated on 100% oxygen can develop lung atelectasis; however, if hyperinflation is performed with an airway pressure of 40 cm H2O and the pressure is maintained for a number of seconds, any atelectasis that has occurred is re-expanded.
The possible physiological side effects of delivered air volume, flow rates, and airway pressure must be carefully considered (Paulus et al. 2012). The most common short term and probably non-relevant side effects are reduction in cardiac output, alteration of heart rate and increase of central venous pressure.
Hyperinflation breaths can also be delivered while the patient is on ventilation. In spontaneously breathing patients ventilator hyperinflation (VHI) is achieved by applying incremental increases in pressure support or volume delivered during controlled ventilation, in order to reach a predetermined target volume. Usually, during the manoeuvre it’s useful to reduce the respiratory rate as well as the inspiratory flow.
Only two studies (Berney and Denehy 2003; Clark et al.1999) have compared MHI and VHI: the techniques were found to be equally effective at clearing pulmonary secretions and improving static pulmonary compliance. Other authors demonstrated that lung hyperinflation reduces the incidence of nosocomial pneumonia in mechanically ventilated patients (Ntoumenopoulos et al. 2002). An advantage of VHI, as opposed to MHI, is less risk of de-recruitment and atelectotrauma. Indeed during VHI, it’s possible to maintain a determined PEEP as the patient is not disconnected from the ventilator.
Recruitment maneuvers (RM) are similar in principle to VHI, and they are used more frequently by medical staff, especially to treat patients who have ARDS. They involve transient elevations in airway pressure applied during mechanical ventilation to open collapsed lung units and increase the number of alveoli participating in tidal ventilation. Because of significant clinical heterogeneity among different clinical trials, the effects of this treatment on clinical outcomes has not been well established. A recent meta-analysis has shown that a ventilation strategy that included RM in ARDS patients reduced ICU mortality without increasing the risk of barotrauma, but it had no effect on 28-day and hospital mortality (Hodgson et al. 2016). However, a recent RCT has disowned these results, showing that in patients with moderate to severe ARDS, a strategy with RM and titrated PEEP compared with low PEEP increased 28-day all-cause mortality (Cavalcanti 2017). In conclusion, nowadays the routine use of lung RM is not recommended.
When applying MHI, VHI, or eventually and in selected cases RM, the clinician needs to be mindful regarding the possible adverse effects of these manoeuvres. The increase in intra-thoracic pressure and subsequent reduction in venous return may lead to a reduction in cardiac output and arterial pressure, compromising cardiovascular stability. The increase in tidal volume may also lead to volutrauma or damage to any recent surgical lung anastomosis. If there are concerns regarding the risk of barotrauma, a manometer can be put into the MHI circuit.
Deep breathing exercises, consisting of a sequence of deep breaths, using the diaphragm rather than accessory muscles of respiration can be an alternative for spontaneously breathing patients. Frequently this type of RP is performed with a device (incentive spirometry), that has the advantage of giving feedback on performance from the gauge. The use of such a device is not considered in the early phase of treatment, as these techniques require substantial co-operation from the patient.
2. Drainage of endobronchial secretions
Adequate and efficient clearance of endobronchial secretions is a crucial point for maintaining airway patency and reducing the number of infections, morbidity, and mortality of critically ill patients. Patients admitted to ICU often suffer from severe impairment of airway clearance capacity (Konrad et al. 1994). Muscular weakness reduces the strength and effectiveness of cough reflex, which in turn makes it more difficult to expel secretions. Moreover, in mechanically ventilated patients, it’s added the impossibility of effectively closing the glottis due to the presence of the intratracheal tube in the proximal airway. Furthermore, the presence of a tracheal tube leads to bacterial translocation from the oropharynx to the lower airways.
Due to the combination of these factors, it’s impossible to generate an expiratory flow sufficient for expulsion of the mucus. In turn retention of mucus represents a factor that determines the deterioration of respiratory conditions, creating a growth medium for bacteria leading to pneumonia and consequent prolongation of MV and length of stay, and increased morbidity and mortality (Sackner et al. 1975). Moreover, patients are often kept in semi-recumbent position in order to prevent aspiration phenomena and the consequent ventilator-associated pneumonia [VAP] (Salamone et al. 2016); however, in this position the trachea is oriented not horizontally but rather obliquely and the gravity force makes the function of mucociliary escalator diminished (Bassi et al. 2008). In addition, conditions such as prolonged entrapment, comorbidity, administration of drugs (especially sedatives, analgesics, and neuromuscular blockers), fluid restriction, and frequent respiratory infections contribute to the overproduction and accumulation of mucus (De Jonghe et al. 2007; Ray et al. 1974). Finally, MV is itself a cause of rapid deterioration of the diaphragm function, the main muscle involved in breathing, and therefore also in mucociliary clearance (Callahan 2008).
As different mechanisms can be responsible for secretions retention, it’s important for clinicians and physiotherapists to identify the problem correctly and select the correct intervention to facilitate sputum clearance. Different therapeutic strategies can be adopted to optimise bronchial hygiene and improve respiratory mechanics, including pharmacological and non-pharmacological treatments. It is unlikely to find a perfect system to clear the airway from the mucus. Generally, airway clearance regimes act through different mechanisms that improve airflow and reduce the viscosity of the mucus, both of which are important factors. Techniques usually exploit the gas-liquid interaction principle between the airflow generated by respiratory acts and the secretions contained within the airways (Benjamin et al. 1989). The mucus, pushed by traction forces generated by the aerial column that moves above it, moves deep into the airways (during the inspiration) or cranial direction (during exhalation). Part of the techniques for secretions drainage is based on the generation of an asymmetrical flow pattern, with an expiratory flow that exceeds the inspiratory flow, thus carrying the mucus outside the airways (Atkinson 2002).
In intubated and mechanically ventilated patients it is always advisable to ensure correct systemic hydration and appropriate airway humidification in order to prevent tenacity of the mucus and consequent adhesion and development of atelectasis. Correct humidification can be obtained via an active or passive humidifier. Contrarily, the routine use of N-Acetyl Cysteine or sodium chloride instillation are not recommended (Atkinson 2002).
In intubated patients with a tendency to retention of secretions, tracheo-broncho-aspiration often represents the terminal phase of the unblocking manoeuvres. When secretions are mobilised from the more peripheral airways centrally, they can be removed via suction. Suctioning can be performed through flexible bronchoscopy or using a catheter. In spontaneously breathing patients the catheter is usually inserted through the nostril, except in case of severe coagulopathy and basal skull fracture, when oro-pharyngeal airways is preferred. Vomiting and laryngospasm may occur if upper airway reflexes are still present. In patients with an artificial airway (endotracheal tube or tracheostomy), suction can be performed via an open or a closed technique. In hypoxaemic patients, suctioning may determine complications, particularly the deterioration of oxygenation and lung derecruitment, which have the potential to worsen lung injury. To prevent or limit these complications, an increased fraction of inspired oxygen (FiO2) should be administered or, when possible, open suctioning should be avoided, and closed systems should be preferentially used (Maggiore and Volpe 2011; Maggiore et al. 2013). Indeed the advantage of a closed circuit is that disruption of the circuit is minimised; however, no clear benefits have been demonstrated in terms of infection reduction.
Mechanical insufflation-exsufflation uses positive pressure to promote maximal lung inflation through a gradual application of positive pressure, followed by a rapid switch to negative pressure. This change produces a high expiratory flow which simulates a powerful cough. The cough assist has proved to be effective in improving clearance in patients with neuromuscular diseases (Gomez-Merino and Bach 2002). Goncalves et al. found that the application of mechanical insufflation-exsufflation as part of an extubation protocol may reduce reintubation rates and ICU length of stay (Vianello et al. 2011; Goncalves et al. 2012; Guérin et al. 2011). Chatwin et al. compared conventional physiotherapy with physiotherapy plus in-exsufflation in patients treated with non-invasive ventilation (NIV); in-exsufflation shortened the treatment time in the ICU without any difference in secretion clearance (Chatwin and Simonds 2009). Despite these benefits, the use of this method is to be reserved only for selected cases among ICU-patients at high risk of severe hypoxaemia and development of atelectasis, since disconnection from the MV and application of negative pressure can aggravate the phenomena of airway collapse. Other main contraindications to this technique are undrained pneumothorax, major cardiovascular instability, and flail segments. Relative contraindications are emphysematous bullae and head-injury.
Postural drainage traditionally includes gravity-assisted positions, deep breathing exercises, chest clapping, shaking or vibration, and incentivised cough. Postural drainage is eventually associated with percussions, a technique that, when performed by a trained operator, has proved to be effective in facilitating the mobilisation of secretions. Percussions are performed using cupped hands to clap the chest wall over the affected part of the lung (Andrews et al. 2013). This combination of techniques was shown to improve lung collapse in mechanically ventilated patients (Chen et al. 2009). As an alternative to percussions, vibrations can be performed manually or using mechanical devices to compress with a high frequency the chest wall. McCarren and colleagues founded that vibrations increased peak expiratory flow rates by more than 50% over relaxed expiration (McCarren et al. 2006). Ntoumenopoulos et al. demonstrated a direct relationship between percussions and vibrations and the reduction in the incidence of VAP by 31% (Ntoumenopoulos et al. 2002).
High-frequency oscillations (HFOs) can be applied either directly to the airway opening (High-Frequency Ventilation, HFV; Intrapulmonary Percussive Ventilation, IPV), or to the surface of the rib cage (High-Frequency Chest Wall Oscillations, HFCWO, also called High-Frequency Chest Wall Compressions, HFCWC). The IPV consists of the application at the opening of the airways of high-frequency ventilation modality that can be superimposed on spontaneous breathing. Oscillations with a frequency typically between 10 and 20 Hz (Freitag et al. 2007) are generated by a compressor and transmitted inside the airway, inducing alveolar recruitment. IPV, therefore, creates a bias between expiratory and inspiratory flow, and in particular generates an expiratory flow that exceeds about 4 times the inspiratory flow. This may reduce respiratory muscle load and help to move airway secretions. A second mechanism through which IPV improves clearance is the modification of the rheological properties of mucus: there is evidence that the application of oscillations within the airways with a frequency between 12 and 22 Hz may decrease the viscosity and thickness of the mucus, thus favouring its mobilisation (Tomkiewicz et al. 1994). Positive effects from this technique have been shown in patients with respiratory distress, neuromuscular diseases, and pulmonary atelectasis (Chatburn 2007; Vargus et al. 2009: Dimassi et al. 2011).
Physiologic effects of IPV were studied by Vargas et al. in intubated patients affected by chronic obstructive pulmonary disease [COPD] (Vargus et al. 2009). IPV promotes secretion clearance (Cioffi et al. 1989), gaseous exchange and it decreases the ICU stay in patients with COPD which undergo NIV (Antonaglia et al. 2006). Dimassi et al. assessed the short-term effects of IPV in patients at high risk for extubation failure who were receiving NIV after being extubated. This study concluded that both NIV and IPV reduced the respiratory rate and work of breathing (Dimassi et al. 2011). Studying a population of tracheostomised patients with prolonged weaning, Clini and colleagues documented that twice a day addition of IPV to regular treatment with conventional physiotherapy (CPT) improves gas exchange and expiratory muscle performance, reduces the incidence of nosocomial pneumonias, although it does not significantly reduce the number of atelectases and the need for toilet broncoscopies (Clini et al. 2006). Although some positive data have emerged about the possible benefits deriving from the use of IPV, to date the value of this method has not yet obtained consistent validations with regard to important long-term outcomes among patient requiring invasive mechanical ventilation (IMV). Therefore, IPV is recommended only for treatment atelectasis and retention of mucus in patients with COPD, cystic fibrosis or postoperative respiratory complications.
HFCWO is an airway clearance technique consisting in external chest wall oscillations applied to the surface of the chest using an inflatable vest that wraps around the chest. The high-frequency pulses and the consequent pressure fluctuations result in slight compressions and releases of the underlying thoracic surface. This produces an oscillatory air flow superimposed to the normal air flow presented by the patient in spontaneous breathing or to the air flow delivered by the ventilator in case of a patient on MV (Hansen and Warwick 1990). The effect of secretions mobilisation is determined through the development of a flow bias between inspiratory and expiratory flow, but also through the modification of the rheological properties of the mucus and the strengthening of the ciliary beating (King et al. 1983; Denton et al. 1968). HFCWO has a wide application among patients affected by cystic fibrosis and bronchiectasis or in patients with neuromuscular disease, but only limited data are available on its use in the critical care settings. In a study conducted by Ndukwu et al. tracheostomised patients were more likely to be weaned from MV when treated with HFCWO vs. conventional physiotherapy [CPT] (Nduko et al. 1999). Moreover, HFCWO has been shown to be effective in improving secretion clearance in a population of adults /elderly patients who have been extubated after prolonged IMV [> 21 days] (Huang et al. 2013).
Despite these positive results, the application of HFCWO did not increase the success rate of weaning after extubation. Nowadays data on the application of HFCWO to patients on IMV are few; to date, only five trials have been published. Comparing HFCWO and CPT (percussion technique and postural drainage) in 9 intubated patients, Whitman and coworkers found no differences in the amount of sputum and in arterial oxygen saturation (SpO2), heart rate and arterial pressure (Whitman et al. 1993). Similarly, Unoki and colleagues did not find significant improvement in clinical outcomes [oxygenation, alveolar ventilation, and removal of secretions] (Unoki et al. 2005). Clinkscale and collaborators applied HFCWO to critically ill patients admitted to a Respiratory Intensive Care Unit and found that, compared to CPT, HFCWO is associated with lower values of pain assessment scores (Clinkscale et al. 2011). The authors found no significant differences between the two treatment groups with regard to the length of stay in ICU or hospital. The major limitation of this study was that only a minority of enrolled subjects was intubated at the time of treatment with HFCWO. More recent trials have shown a positive impact of the technique on clinical outcomes. The results of the Taiwanese RCT enrolling patients intubated because of severe acute respiratory failure secondary to pneumonia showed that HFCWO produces short-term adverse effects on SpO2, while not significantly altering Ppeak, minute ventilation, systolic pressure and heart rate (Chuang et al. 2017). In all patients, the mobilisation of endobronchial secretions was improved, but did not reach a statistically significant difference with respect to CPT. HFCWO is generally well tolerated, and the authors of the latter trial didn’t detect significant changes in cardiorespiratory parameters. Similarly, Gugliotta et al. obtained a positive effect on secretion removal in invasively ventilated patients, as confirmed by changes in end-expiratory lung impedance [Deeli] (Gugliotta et al. 2016). Despite these promising results, to date, no large RCTs have been carried out to systematically analyse the possible beneficial effects or adverse events of HFCWO on the cardiorespiratory system. As a result, the applicability and safety of this physiotherapeutic technique among critically ill patients is still unknown, as well as its effectiveness.
Positive expiratory pressure (PEP) consists of a one-way valve through a mask or a mouthpiece connected to an adjustable expiratory resistor to enhance and promote secretion removal by stenting airways, increasing intrathoracic pressure, or increasing functional residual capacity (Myers 2007). The possible benefits of this technique are still under investigation (Örman and Westerdahl 2019), and its application to ICU patients is still to be studied.
During last decades there has been increasing involvement of physiotherapists in the assessment and setting up of non-invasive ventilation (NIV), as the aims of respiratory physiotherapy and NIV frequently overlap (Ntoumenopoulos et al. 2011). Bilevel Positive Airway Pressure (BiPAP) is used predominantly for patients with exacerbations of COPD. However, there is an increasing application to patients who are at risk of post-extubation respiratory failure and also as part of a weaning strategy for patients with hypercapnic respiratory failure (Gosselink et al. 2008). Continuous Positive Airway Pressure (CPAP), BiPAP, or both are used for patients with acute cardiogenic pulmonary oedema and can also be used for selected patients with acute hypoxaemic respiratory failure from other aetiologies. NIV is known to decreased work of breathing and increase dynamic lung compliance, tidal volume, and inspiratory capacity with a subsequent improvement in blood gases. NIV can be a valuable complement to RP, which can be performed preferably during NIV sessions with a potential increase in patient’s tolerance to efforts required by physiotherapy, in patient’s co-operation. Moreover, the manipulation of the ventilator setting may facilitate the mobilisation and expulsion of secretions with potential improvement in lung mechanics and secretion expectoration. The strategies that can be adopted in intubated patient consist in generating lower inspiratory flows that translates into an increase in the difference in expiratory-inspiratory flows and categorically in the improvement of the transport of mucus. However, although there are encouraging results emerged from laboratory studies, the confirmations of this hypothesis in vivo are still few, and the application of NIV in order to facilitate secretion removal remains rare.
Another promising therapeutic option for critically ill patients is represented by high flow nasal cannula (HFNC). Since its introduction, HFNC has been applied to treat patients with hypoxaemic respiratory failure and to prevent reintubation in patients at risk of extubation failure (Maggiore et al. 2014; Hernández et al. 2016). In these patients, compared with conventional O2 therapy, HFNC improves oxygenation, ameliorates the washout of pharyngeal dead space, reduces airway resistance, increases end-expiratory lung volume, and generation of positive airway pressure and decreases the work of breathing [WOB] (De Mussi et al. 2018). Moreover, HFNC is associated with greater comfort and tolerance, lower dyspnoea, lower dryness of upper airways and lower desiccation of secretions, compared to standard oxygen (Tiruvoipati et al. 2009; Rittayamai et al. 2013; Williams et al. 1996).
3. Mobilisation
During an acute phase of critical illness, the patient is usually confined in bed and, due to sedatives, he may not be collaborative. It’s well known that bed rest is a major problem associated with skeletal muscle weakness, disuse atrophy, and deconditioning (Stevens et al. 2007; Hermans et al. 2014). Drugs such as steroids and neuromuscular blockers exacerbate this critical illness neuromyopathy and malnutrition further compound this problem.
Indeed, early mobilisation of patients leads to improvements in peripheral and respiratory muscle strength, and, among patients treated with MV, it results in greater ventilation-free time (Wang et al. 2007). Mobilisation activates muscle metabolism and optimises oxygen transport by enhancing alveolar ventilation and ventilation/perfusion (V/Q) matching; it also represents a gravitational stimulus to maintain or restore circulation and normal fluid distribution in the body (Stiller et al. 2004; Dean 1994). Early mobilisation can be performed also in unconscious or sedated patients (Hanekom et al. 2011), and algorithms and protocols have been proposed as a guide in selecting eligible patients for mobilisation and providing appropriate treatment strategies (Liu et al. 2018). However, to date, there is insufficient evidence on the effects of early mobilisation of critically ill patients on physical function or performance, adverse events, muscle strength and health-related quality of life. This low-quality evidence is mostly due to small sample sizes, great variation, and inhomogeneities in the interventions and outcomes used to measure their effect. Nevertheless, the European Society of Intensive Care Medicine statement (Gosselink et al. 2008) suggests that active or passive mobilisation and muscle training should be instituted as soon as possible.
One of the interventions most frequently applied in the critical area is body positioning (Ambrosino et al. 2012). The physiological rationale for changing patient position is that it prevents dependent airway closure and atelectasis, pooling and stagnation of pulmonary secretions, and subsequent infection that may result from prolonged immobility (Raoof et al. 1999). In the supine position, lung volumes in dependent regions may reduce above functional residual capacity, leading to airway closure and atelectasis. At the same time airway resistance increases, mostly resulting from abdominal and chest wall compression, and this hinders the expulsion of secretions. In addition, the supine position, compared with the head-up position, has been found as a potential risk factor for pulmonary aspiration of gastric contents in patients receiving MV (Torres et al. 1992). Moreover, the semirecumbent or upright position (bed head positioned at 45°) is able to prevent pulmonary aspiration, to improve V/Q matching, lung volumes and mucociliary clearance, to reduce the respiratory rate, the WOB and the work of the heart, and to increase tidal volume and inspiratory flow rate (Gosselink et al. 2008; Zafiropoulos et al. 2004). Prone positioning has become an important instrument for the treatment of patients with ARDS, because it improves V/Q matching and oxygenation, redistributes oedema, and increases functional residual capacity (Jolliet et al. 1998; Mure et al. 1997). For patients with unilateral lung disease, placing the affected lung uppermost results in facilitating drainage from lung segments and improvement of V/Q matching.
Continuous lateral rotational therapy (CLRT) can be obtained through the use of specialised beds that rotate along the longitudinal axis, up to an angle of 60° onto each side, with a pre-set speed and degree of rotation (Traver et al. 1995). It has been hypothesised that CLRT can reduce the risk of sequential airway closure and pulmonary atelectasis, resulting in a reduction of lower respiratory tract infection and pneumonia, duration of endotracheal intubation and length of hospital stay (Ambrosino et al. 2012; Kirschenbaum et al. 2002). However, this therapy is not commonplace in the ICUs due to a lack of solid evidence of its cost-effectiveness. Adverse events are not common during positioning; however, as previously said, it’s best to select the patient to treat considering individual specific risks and benefits of the process. Another important aspect to be considered is that prolonged immobility can determine joint contractures that limit the possibilities for future mobilisation. For patients who are unconscious passive limb exercises, such as stretching, splinting (Gosselink et al. 2008) or passive movements with continuous passive motion (CPM), should be applied daily as early as possible (Reid and McNair 2004; Griffiths et al. 2011). For patients who are conscious and able to follow instructions specific active therapy modalities involve any of the following: active turning and moving in bed, active-assisted and active exercises, pedal cycles in bed, tilt table, sitting at the edge of the bed, getting out of bed via mechanical lifting machines standing, transfers from bed to chair, chair-based exercises, and walking [with or without the help of standing or walking aids] (Sommers et al. 2015).
In conclusion, although there is unequivocal evidence that prolonged bed rest results in deconditioning, there is no published study, in patients receiving MV in ICU, investigating the effect of mobilisation on the pulmonary function, the weaning process, or the length of stay. However, the combination of multiple techniques involving mobilisation may result in the best clinical outcomes for patients admitted to an ICU (Ntoumenopoulos et al. 2002).
4. Muscular training
As previously said, critical patients are often forced to long periods of bed rest, with consequent progressive deconditioning and establishment of generalised muscle weakness. ICUAW is frequently observed in a substantial proportion of ICU-patients (Puthucheary et al. 2013; Nordon-Craft et al. 2012; Mendez-Tellez and Needham 2012). The prevalence of ICUAW is 25–40% in patients ventilated for ≥ 48 h and even higher in patients with sepsis or a prolonged ICU length of stay (LOS) (Appleton et al. 2015; De Jonghe et al. 1998; Tennilä et al. 2000). Muscle wasting develops since the very first week of illness, with more severity in patients with multiorgan failure compared with those with single organ failure (Puthucheary et al. 2013). ICUAW, including critical illness polyneuropathy, critical illness myopathy, and critical illness polyneuropathy and myopathy, is characterised by a profound weakness that is greater than normally expected from prolonged bed rest, and is therefore defined as clinically detected weakness in critically ill patients in whom there is no plausible aetiology other than critical illness (Nordon-Craft et al. 2012; Hermans and Van den Berghe 2015; Kress and Hall 2014). This deficit in almost all cases involves skeletal muscles, but the respiratory muscles and diaphragm may also be profoundly altered. ICUAW limits the activities of daily living, and delays rehabilitation and recovery (Fan 2012; Herridge et al. 2011). Its possible aetiologies are mainly represented by deconditioning, disuse atrophy, systemic inflammatory response syndrome, sepsis, and multiple organ dysfunction syndrome, hyperglycaemia and medications [corticosteroids and neuromuscular blocking agents] (Hermans et al. 2014; Stevens et al. 2009). MV itself may adversely affect the diaphragm's structure and function, with a process termed ventilator-induced diaphragmatic dysfunction (Petrof et al. 2010). Indeed, patients who undergo prolonged periods of MV demonstrate a decrease in respiratory muscle endurance and are at risk of respiratory muscle fatigue (Chang et al. 2005).
Respiratory muscle weakness and, in particular, the imbalance between the muscle strength and load upon the respiratory system is one of the major determinants of weaning failure. However, the rationale of respiratory muscle training (RMT) in ICU is controversial, since little physiopathological information is available to support the specific use of respiratory muscles training among critical care patients. RMT has been associated with favourable weaning in ICU ventilatory-dependent COPD patients (Aldrich et al. 1989; Aldrich and Uhrlass 1987, Martin et al. 2002). The technique applied in these studies, Inspiratory muscle training (IMT), targets the muscles of inspiration, namely the diaphragm and accessory inspiratory muscles, with the aim of increasing inspiratory muscle strength and endurance. In ventilated patients, the IMT device is incorporated into the ventilator circuit with an adaptor or connector, and IMT can be undertaken in several ways: isocapnic/normocapnic hyperpnoea training, resistive flow training, threshold pressure training, or adjustment of the ventilator to provide a training load for the inspiratory muscles. Since the results on the application of this technique in patients on IMV are controversial (Caruso et al. 2005), its routine use is not common.
With regard to peripheral muscles, during a period of inactivity, muscle mass declines and its efficiency to perform aerobic exercise is reduced. The effects of deconditioning are higher during the first week of immobilisation, when loss of strength reaches 40% (Bloomfield 1997). Muscular atrophy induced by bed rest is made worse by the association of critical illness polyneuropathy and malnutrition with protein wastage and bone demineralisation. Passive and active training of skeletal muscles in ICU should be aimed at restoring a muscle strength that allows basic daily life-activities and the ability to walk independently. For patients in critical conditions that cannot cooperate, neuromuscular electrical stimulation (NMES) is used to improve muscle performance. This therapy creates a passive contraction of skeletal muscles through the use of a low-voltage electrical impulse delivered through electrodes placed on the skin over the target muscle groups. The low-volt stimulation activates motor nerves and mimics the effects of muscle contractions during mild exercise, with improvement in intramuscular blood flow, maximal muscle force output, and force endurance. Moreover, the repetitive contraction of muscle fibres helps in maintaining muscle tone. This passive training is generally well tolerated and was shown to significantly improve muscle strength and respiratory rate and decrease the number of days needed to transfer the patients from bed to chair among COPD patients receiving MV (Zanotti et al. 2003; Meesan et al. 2010; Williams and Flynn 2014). More recently the effects of NMES were studied with muscle ultrasounds in 26 critically ill patients; Gerovasilli et colleagues confirmed that this intervention is well tolerated and found out that it may help in preserving the muscle mass (Gerovasili et al. 2009). However, among critically ill patients the efficacy of NMES may be reduced, especially in patients having sepsis or oedema, or receiving vasopressors because these conditions might impede with the generation of a sufficient muscle contraction by electrical muscle stimulation (Segers et al. 2014; Fossat et al. 2018).
For patients in a more stable condition, the sessions of muscular training usually involve passive or active movements of lower and upper extremities, eventually enhanced by lifting light weights or pushing against resistance. Rehabilitation-related technologies, such as cycle ergometer (CE), may play an important role in improving muscle strength and physical function in ICU patients. With this stationary cycling apparatus, patients can exercise through passive, active-assisted, or active training. Among sedated, immobile patients with severe critical illness CE may help in preserving muscle architecture (Griffiths 1997; Burtin et al. 2009). However, despite its potential benefits, rigorous evaluation of CE as a rehabilitation therapy for ICU-patients is still limited.
Later, as soon as the patient is able to stand, the physiotherapist should start progressive walking retraining, aided by a rolling walker if needed. Strategies include patient mobilisation based on a progressive sequence of activities like decubitus change and functional positioning, cycling and sitting in bed, and standing, static walking, transferring from bed to chair, and walking. Oxygen therapy or a portable NIV-respirator can eventually be used to decrease the WOB during walking. In a controlled trial, Nava et al. showed that the application of a step-by-step peripheral muscle retraining program to COPD patients recovering from an acute episode of hypercapnic respiratory failure and admitted in ICU has been associated with improvement in the patient’s exercise capacity and symptoms score as compared to controls (Nava 1998).
Conclusions
Patients who survive hospitalisation in ICU are at increased risk for reporting negative complications of critical illness, even after stabilisation of the conditions. They frequently develop neuromuscular, cognitive and psychosocial deficits which can result in decreased quality of life, impaired ability to function autonomously and altered occupational performance. This significant morbidity represents a burden to patients, caregivers, and society.
In recent years the profile of rehabilitation in critical illness to mitigate these long-lasting complications has increased, and respiratory rehabilitation has become a cornerstone in the comprehensive management of ICU-patients. There is growing evidence that an early and progressive programme is feasible with low risk to the patient. Respiratory physiotherapy determines recognised short-term benefits, preventing some ICU complications, such as PICS and ICUAW, and preserving or recovering patients’ functionality. The treatment strategies should be applied in the early phase of illness, as soon as the patient reaches cardiorespiratory and neurological stabilisation. Even sedated and not collaborative patients can be passively treated. Respiratory physiotherapy programmes are usually based in four areas: lung hyperinflation, drainage of endobronchial secretions, mobilisation and muscle training. Benefits potentially associated with respiratory physiotherapy include shortening of the weaning time, restoration of functional capacity and physical independence, reduction of length of stay and healthcare costs. However, no significant effects have been shown on cognitive function or mortality.
A multidisciplinary team approach (physical therapist, physician, nurse) is fundamental to conduct a safe and effective rehabilitation programme.
With a growing population of ICU survivors, greater standardisation of respiratory physiotherapy techniques is needed, especially given the unique challenges presented by ICU patients and the environment. There is great potential for exploring the benefits of interventions, and we encourage for large scale, rigorous randomised trials to further explore long-term outcomes and the ideal dose and timing and the effect of specific techniques on specific conditions. Furthermore, we need to improve ICU organisation and teams to be able to deliver early intervention.
























