ICU Management & Practice, Volume 20 - Issue 4, 2020
Critically ill patients can develop a series of complications due to ICU risk factors that may lead to permanent disability. The implementation of strategies to reduce its incidence is essential.
Nowadays, the possibilities of survival of critically ill patients have grown due to technological and medical advances. This leads to a series of consequences owing to the critical pathology itself, medical procedures and the length of stay in the Intensive Care Unit (ICU). Many functional and physical impairments can be established during this critical time and remain even beyond hospital discharge. Furthermore, cognitive performance, psychological status and quality of life (QoL) can be compromised not only for the patient but for their family as well. A build-up effect can be generated if any of the impairments affect one or some of these individuals, starting from a disorder in a micro-state of the individual climbing up to an insult in their social role, establishing a disability that could not only affect the individual’s functionality but the whole society as well (Van Zanten et al. 2013; Needham et al. 2012; Elliot et al. 2014).
Different modifiable and non-modifiable factors can influence the functional outcome of the critically ill patient. Modifiable factors and relatively simple interventions can be taken into consideration inside the ICU, focused on preventing functional complications. Some of these are early mobilisation (EM), reduced sedation, non-pharmacological anti-delirium measures and the empowerment of the family in patient care. Age, gender, previous functionality, chronic diseases, sarcopenia and fragility before hospital admission are some non-modifiable situations that can impact directly in the functional prognosis. Hence, the best way of predicting the future of the patient is by thoroughly and comprehensively knowing the individual’s past. Complications that culminate into a disability should be considered in conjunct as Post-Intensive Care Syndrome (PICS) (Elliot et al. 2014).
The conceptualisation of PICS involves three specific domains: physical, cognitive and mental health related (psychological) functions. Any affection in these domains obtained in the ICU that endure through hospital discharge in the patient and its family (PICS-F) should be considered part of this syndrome, including paediatric population (PICS-p). Alterations after a patient’s death are considered PICS-F. These impairments caused by PICS can perdure years after hospital discharge and an appropriate follow-up is needed in order to minimise the impact and presence of any disability (Harvey and Davidson 2016).
Post-Intensive Care Syndrome
PICS can affect the patient in the three domains mentioned above: physical, cognitive and mental health related (Elliot et al. 2014; Harvey and Davidson 2016).
Physical Impairment
The majority of the physical sequelae derives from ICU-acquired weakness (ICU-AW). ICU-AW is defined as a neuromuscular dysfunction alongside symmetrical and progressive muscular strength loss without any cause other than the admission into the ICU. It is clinically diagnosed with the Medical Research Council Sumscore (MRC-SS) of ≤ 48, or by the measurement of grip strength (< 7 kg in women and < 11 kg in men). The aetiology can be due to critical illness myopathy, critical illness polyneuropathy or a unity of both, known as critical illness polyneuromyopathy. Owing to the hypermetabolic state through which the critically ill patient undergoes, muscle mass is susceptible to decline drastically. This is associated with poorer outcomes. Therefore, nutrition and exercise are fundamental keys towards the preservation of muscular health (Turan et al. 2020; Farhan et al. 2016; Hermans and Van den Berghe 2015; Ali et al. 2008).
The incidence of ICU-AW oscillates between 25-100% resulting in longer lengths of stay inside the ICU and hospital, weaning failure due to its relation with diaphragm dysfunction, and increase in mobility problems and mortality. Physical sequelae, including ICU-AW, occur in 25-80% of individuals who undergo invasive mechanical ventilation (IMV) (Harvey and Davidson 2016, Vanhorebeek et al. 2020; Kress and Hall 2014; Desai et al. 2011). ICU-AW is much more complex than muscular atrophy secondary to immobility. It is a muscular disorder that compromises the excitability, quality, and mitochondrial function of the myocyte. This could lead to more severe complications such as muscle infiltrations and necrosis (Kress and Hall 2014; Sandri 2013). Alterations in functionality that come from ICU-AW can perdure years after hospital discharge, affecting directly the QoL and the possibility of reincorporation into their previous activities (Kress and Hall 2014; Formenti et al. 2019; Hopkins et al. 2017).
On the other hand, a deterioration in the resistance to physical exercise in patients undergoing critical illness has been found within 6 to 12 months after ICU discharge, characterised by a decrease in the metres travelled during the 6-minute walk test. Also, the patient can go through a decrease in respiratory function, especially those who have been treated for acute respiratory distress syndrome (ARDS) and in whom the pulmonary parenchyma is damaged permanently. Diaphragm dysfunction also may have a negative impact in resistance to physical exercise. Some patients have been found with a reduction in maximum inspiratory pressure (MIP) up to 15% even 12 months after discharge. Altogether, this generates a decrease in aerobic capacity that can influence the possibility to achieve a successful performance in basic life activities (Hopkins et al. 2017; Herridge et al. 2016).
Additional physical alterations have been reported and involve aesthetic and osteoarticular disturbances. Some of these include stiffness, pain, dental loss, frozen shoulder, skin damage by fluid overload, surgical scars, burns, damage due to endotracheal intubation, IMV, or oxygenation therapy such as post-extubation dysphagia and swallowing and phonation disorders that can have adverse emotional and social impact on the patient (Herridge et al. 2016; Beduneau et al. 2020).
Cognitive Impairment
Cognitive impairment occurs in 30-80% of patients admitted to the ICU. A decrease in correct chore execution, attention span, information processing, problem resolution and accurate perception in location and object position have been observed. Changes in neurological structures have been described among ICU survivors and have been associated with cognitive impairment and delirium. Lateral ventricle enlargement, brain atrophy in frontal lobes and hippocampus, altered white matter, corpus callosum and internal capsule are frequently related (Harvey and Davidson 2016; Desai et al. 2011; Hopkins et al. 2017; Herridge et al. 2016; Briegel et al. 2013; Brummel et al. 2015; Fernández-Gonzalo et al. 2020; LaBuzetta et al. 2019; Ohtake et al. 2018; Inoue et al. 2019; Haines et al. 2015; Davidson et al. 2013).
Delirium
Delirium is a common acute brain dysfunction that affects critically ill patients. Even though delirium was first described 50 years ago, it still remains an underdiagnosed condition in the ICU (Engel and Romano 1959). Evidence has been described in order to prevent, manage or treat delirium in important programmes such as the Society of Critical Care Medicine’s (SCCM) 2018 Clinical Practice Guidelines for the Prevention and Management of Pain, Agitation/Sedation, Delirium, Immobility and Sleep Disruption in Adults Patients in the ICU (PADIS) and the ICU Liberation (ABCDEF) Bundle (Barr et al. 2013; Vasilevskis et al. 2018). In both of these, EM takes an important role to overpower many of the consequences that critical illness comes with, such as delirium. Delirium prevention and management is stated to need a multidisciplinary approach with routine delirium assessment with validated tools and a standardised EM process that is intimately related with minimising sedation, which means awakening and spontaneous breathing trials need to be in coordination (Trogrlić et al. 2015).
Delirium has been associated with an increase in morbidity, mortality, length of stay in the ICU and long-term cognitive impairment (Vasilevskis et al. 2018). Therefore, the need to assess and identify it is evident. The most common method to diagnose delirium in the ICU is the Confusion Assessment Method for Intensive Care Unit (CAM-ICU) (Salluh et al. 2009), with recent studies supporting its use over other ways of diagnosis. Delirium rates are varied depending on the population that is studied and over the interaction of other 100 risk factors described in the literature. A meta-analysis that included 16,595 patients going through a critical illness showed a delirium rate of 31.8% (LaBuzetta et al. 2019; Salluh et al. 2015; Arias-Fernández et al. 2018; Denehy et al. 2017).
The implementation of the correct measures is important for the prevention of delirium much like the decrease in sedation (specially with the use of benzodiazepines), analgesic optimisation in the presence of pain (supporting non-pharmacological measures), considering physiological sleeping time (avoiding nocturnal procedures), allowing sunlight exposure, avoiding restraints or any movement restriction and establishing effective communication channels between patient-staff and patient-family (Inoue et al. 2019; Pandharipande et al. 2010; Smonig et al. 2019; Devlin et al. 2018).
Mental Health-Related Impairments
Psychological consequences include anxiety, depression and sleeping disorders that can persist for months or years. Approximately 10-50% of patients undergoing clinical illness manifest post-traumatic stress disorder (PTSD), persisting up to eight years. These conditions do not only involve the patient but their family as well, during and after clinical illness or death (Harvey and Davidson 2016, Herridge et al. 2016; LaBuzetta et al. 2019; Inoue et al. 2019; Arias-Fernández et al. 2018, Wintermann et al. 2015; Parker et al. 2015).
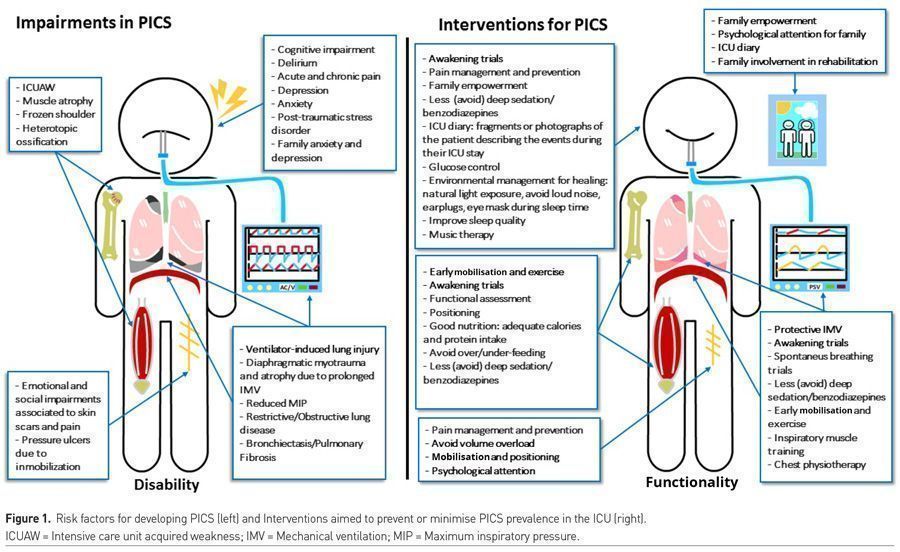
Disability
The mobility level and participation in daily life activities (DLA) and daily life instrumented activities (DLIA) can suffer alterations after critical illness and, in some cases, patients may not be able to reach their functionality level prior to the ICU. A year after critical illness, 33% and 5% of the patients still have problems in at least one DLA or DLIA, respectively. Returning to previous occupation can also be compromised. Three months after ICU, only one third of the patients will return to their jobs and half of this population will return a year after discharge. Survivors of ARDS report an important decline in QoL and functionality up to two years after ICU - manifested by physical exercise intolerance, inability to work or depression (Herridge et al. 2016; LaBuzetta et al. 2019; Inoue et al. 2019; Devlin et al. 2018)
PICS is proven to have a high impact on functionality, QoL, survival and even in economic status among patients and family members overcoming critical illness. This highlights two main necessities: 1) Prevention, and 2) Lowering risk factors associated to PICS.
PICS Prevention Strategies
Identifying symptoms related to PICS can be challenging because of the lack of clinical follow-up between ICU stay, discharge, and home care. Hence, staff in charge of the critically ill patient must be aware of signs associated with PICS.
Prolonged immobility, days undergoing IMV, days inside ICU, sepsis, ARDS, hyperglycaemia, inflammation, hypoxia, electrolyte disorders, malnutrition, dysregulated opioid use, sedatives and neuromuscular blockers are all risk factors associated with PICS in any of its domains. In addition, the effectiveness in PICS management once it is identified tends to be, in the best case scenario, modest. This is why, the most effective way to defeat PICS, is by preventing and minimising its risk factors from day one (Herridge et al. 2016; Inoue et al. 2019; Pandharipande et al. 2010; Devlin et al. 2018; Wischmeyer and San-Millan 2015).
Up until today, the ICU Liberation (ABCDEF + GH) bundle proposed by the SCCM is one of the best strategies in PICS management. This bundle evaluates risks resulting from deep sedation, delirium and immobility (Inoue et al. 2019, Pandharipande et al. 2010, Devlin et al. 2018; Wischmeyer and San-Millan 2015). ABCDEF-GH stands for:
A. Assessment, prevention, management of pain;
B. Both spontaneous awakening and breathing trials;
C. Choice of analgesia and sedation;
D. Delirium assessment, prevention, management;
E. Early mobility and exercise;
F. Family engagement and empowerment; follow-up referrals and functional reconciliation;
G. Good handoff communication; and
H. Handout materials on PICS and PICS-F.
International guidelines provide the interdisciplinary and multidisciplinary teams a set of considerations, recommendations and tools needed for adequate identification and management of PICS (LaBuzetta et al. 2019; Inoue et al. 2019; Pandharipande et al. 2010; Devlin et al. 2018; Devlin et al. 2020). This set is stated in Table 1 and schematically compared with risk factors in Figure 1.
Early Mobilisation - A Cornerstone Against PICS
Certainly, a key intervention in preventing and treating PICS is EM. EM refers to the “E” in the ABCDEF Bundle.
EM and exercise is a strategy used worldwide for the prevention of physical impairments during the ICU stay, among which, ICU-AW, pressure sores and deep vein thrombosis stand out (Wang et al. 2019). Active participation in exercise (referred to as movement generated by the patient) is preferable over passive movement (movement executed by complete assistance, with no voluntary effort by the patient) since it influences reduction in sedation, analgesia optimisation and humanisation of services inside the ICU (LaBuzetta et al. 2019; Inoue et al. 2019; Pandharipande et al. 2010; Devlin et al. 2018; Devlin et al. 2020; Martinez et al. 2020).
EM should start between the second and fifth day of critical illness (Cameron et al. 2015). The implementation of EM protocols based on functional objectives provides a different point of view in the management of the critically ill patient, guiding interventions for the prevention of disability after discharge. Muscular strength assessment through MRC-SS, grip strength or ultrasonography (qualitative and quantitative, like Heckmatt scale or muscular diametre) should be included in such interventions. Nevertheless, in order to achieve an integral approach, functional assessment will expose the true muscular status (Turan et al. 2020; Formenti et al. 2019; Parry et al. 2017; Annetta et al. 2017).
Broad spectrum of functional scales to assess the critically ill have been designed and can guide mobility in a dynamic perspective. It is worth mentioning that the tool selection to identify and diagnose functional status depends on the needs of each service, time availability, and human and material resources at disposition (Parry et al. 2017).
Among the most recognised stand Chelsea Critical Care Assessment Tool (CPAx-Tool), ICU Mobility Scale (IMS), Perme Score, Physical Function in Intensive care Test (PFIT), Functional Status Score for the ICU (FSS-ICU), and others. Functional assessment will individualise the management and highlight opportunities for physical therapy (PT) personnel to prioritise. PT is crucial in the prevention of many of the impairments so far mentioned. One of the major tasks for experts in human movement and functionality is to prioritise the prevention of functional alterations. This is essential considering the association with higher care costs, non-reincorporation to labour activities and even death (Martinez et al. 2020; Parry et al. 2017; Saladin and Voight 2017; Sahrmann 2017).
Implementing an EM programme will assure the development of patient necessity based management and promote the functional preservation of the individual. The ultimate goal is to achieve a positive impact in QoL, socioeconomic status and family wellbeing (Hodgson et al. 2018;, Escalon et al. 2020).
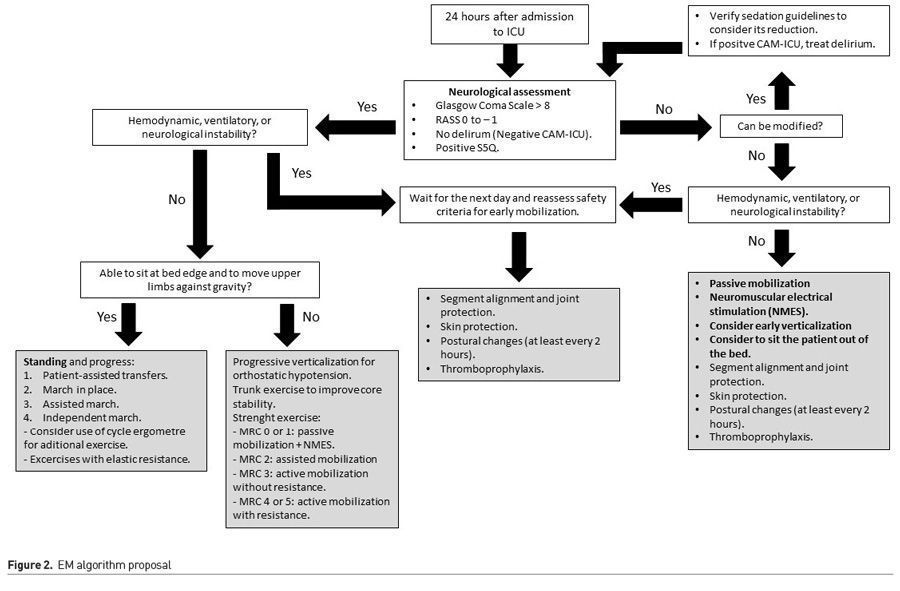
For this purpose, we vastly recommend the implementation of EM programmes as a daily and common practice. An algorithm for the initiation of an EM programme, developed by our team, is illustrated in Figure 2.
Additionally,implementing EM programmes promotes the constant evaluation in sedation, analgesia and delirium. These evaluations can be made through known scales such as Richmond Agitation Sedation Scale (RASS), Confusion Assessment Method for the ICU (CAM-ICU) and Standardized 5 Questions (S5Q). Institutional EM programmes encourage ICU multidisciplinary teams to work towards functional objectives, decreasing the impact of immobility and consider the removal of unnecessary equipment, which is also a risk factor in the development of delirium and a barrier for EM (Hodgson et al. 2018; Zang et al. 2019).
Conclusion
Identifying, preventing and management of long term complications of critical illness is now part of the daily activities for health care providers. Paradigm change in management and objectives inside the ICU, and improvement in the awareness of PICS relies on the whole multidisciplinary team attending critically ill patients. Minimising risk factors associated with PICS requires effective communication, role and capacity recognition, and an understanding of the importance of these during the ICU stay. The correct use of these concepts allows the ICU environment itself to be perceived as an open space and where dialogues are encouraged for the benefit of the centre pieces in this puzzle - the paitents and their families.
Conflict of Interest
None.
References:
Ali NA, O’Brien JM Jr, Hoffmann SP et al. (2008) Acquired weakness, handgrip strength, and mortality in critically ill patients. Am J Respir Crit Care Med, 178(3):261–8.
Barr J, Fraser GL, Puntillo K et al. (2013) Clinical practice guidelines for the management of pain, agitation, and delirium in adult patients in the intensive care unit. Crit Care Med, 41:263–306.
Beduneau G, Souday V, Richard JC et al. (2020) Persistent swallowing disorders after extubation in mechanically ventilated patients in ICU: a two-center prospective study. Ann. Intensive Care 10, 138.
Briegel I, Dolch M, Irlbeck M, et al. (2013) Quality of results of therapy of acute respiratory failure: Changes over a period of two decades]. Anaesthesist, 62:261–270.
Brummel NE, Balas MC, Morandi A et al. (2015) Understanding and reducing disability in older adults following critical illness. Crit Care Med, 43:1265–1275.
For full references please email [email protected],
























