ICU Management & Practice, Volume 22 - Issue 1, 2022
Adverse events (AEs) are common among critically ill patients. Evidence about the nature, preventability and predictability of AEs can be used to reinvest in efforts to reduce them and improve patient safety in ICUs.
Case
Mr. A, a previously well 70-year-old male was admitted to the intensive care unit (ICU) with respiratory failure secondary to novel SARS-CoV-2 (COVID-19). He received two weeks of aggressive therapies before his clinical condition improved, had a tracheostomy and was successfully liberated from mechanical ventilation. On day 16, with the ICU at full capacity, he had a clogged nasogastric tube replaced before being transferred to a medical unit. Twelve hours later he was readmitted to the ICU with respiratory failure and copious tube feed-coloured secretions were aspirated from his tracheostomy.
Why did this adverse event (AE) occur? What could have been done to prevent this AE? How should the medical team proceed?
Safety in the ICU
Critically ill patients in ICUs are the most vulnerable patients within the healthcare system. Their critical illness and complex care puts them at risk for AE - unintended negative consequences of healthcare delivery that compromise patient safety (Kohn et al. 2000).
The estimated rate of AEs in critically ill patients ranges widely from 15% to 51% of ICU patients, with considerable variability between studies (Ahmed et al. 2015). The reason for this variation is not well understood but likely is related to patient-level and study-level factors (Sauro et al. 2021). Nevertheless, it is clear that the number of critically ill patients that experience AEs is higher than that of the general hospital population. While an estimated 8% of hospital patients experience an AE (Sauro et al. 2015; Brown et al. 2004; Brennan et al. 1991), most estimates of AEs among critically ill patients are at least twice as high. What drives this large difference and what can be done to reduce AEs and improve safety among ICU patients? The objective of this study is to understand safety in the ICU and explore evolving trends in AEs in ICUs.
Methods
We conducted a sub-analysis of a previous systematic review and meta-analysis of hospital AEs (Sauro et al. 2021) and augmented the systematic review with a narrative review of more recent studies. We included 11 studies that provided estimates of AEs in the ICU from the previous systematic review (from inception of the databases until 2017) and augmented this search with literature examining AEs in ICU from January 2017 until present (October 2021). We searched Medline (OVID) using terms from Sauro et al. (2021) (previous systematic review) and Ahmed et al. (2015) (systematic review of AEs in ICU) using MeSH terms, text words and synonyms related to adverse events and ICU (Appendix A).

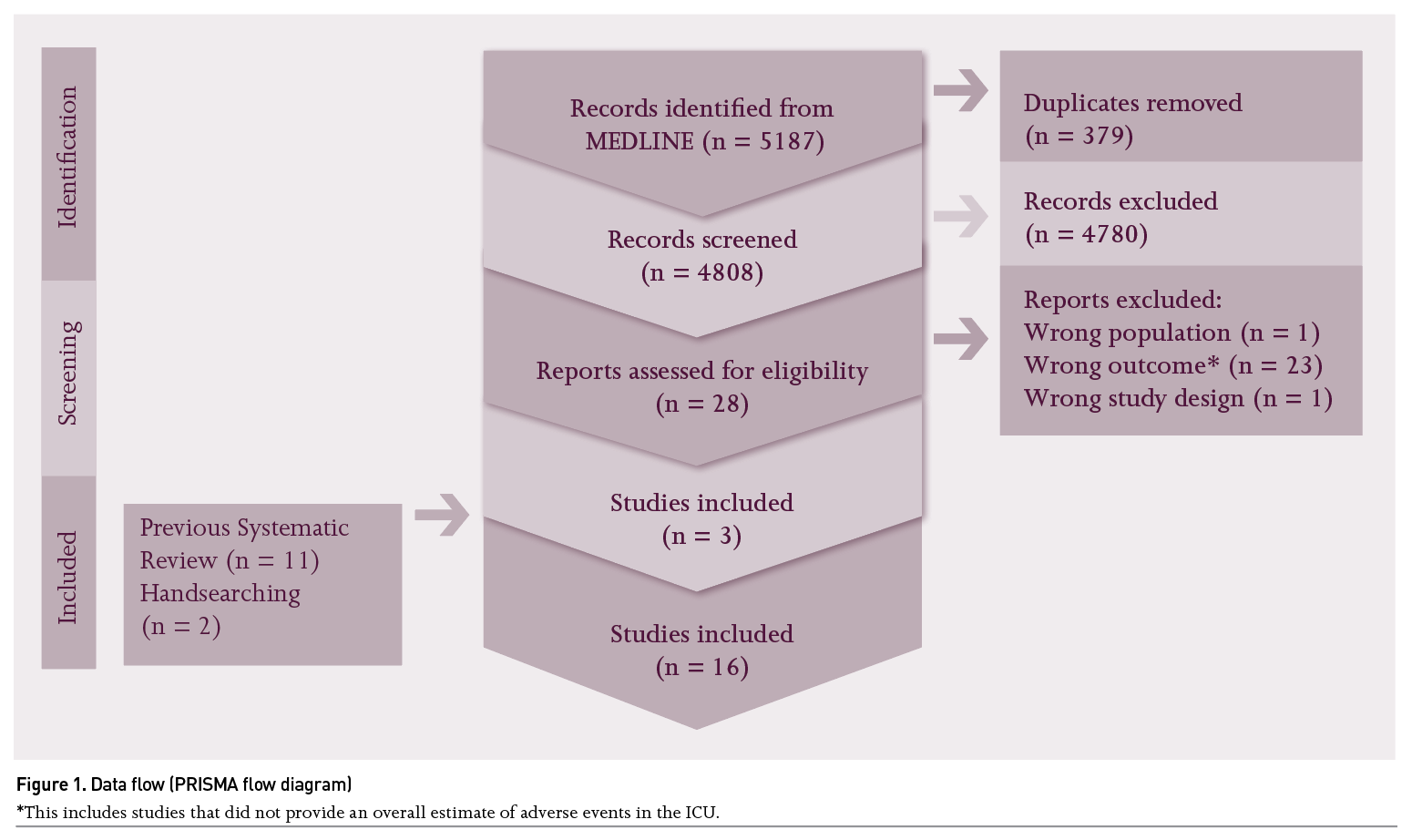
The new search yielded 4808 non-duplicate references. After screening titles, abstracts, and full-texts three studies were included from the new search. Two additional articles were identified through handsearching reference lists of included articles. In total 16 studies (11 identified from Sauro et al. (2021)
plus five identified from the updated search) were included in this review (Figure 1).
How Common are AEs in the ICU?
Nearly a quarter (24.9%, 95% CI=16.4, 33.3, number of studies [n]=15) of critically ill patients experience at least one AE during their ICU stay, at a rate of 8.5 AEs per 100 patient days (95% CI=6.24, 10.74, n=8). Given that AEs occur more commonly among those who are older, have multimorbidity and more severe illness (Sauro et al. 2021; Sauro et al. 2020a; Zegers et al. 2011; Sauro et al. 2017), it is not surprising that critically ill patients are more likely to experience an AE than the general hospital population, which has been reported to be 8% (Sauro et al. 2021; Baker et al. 2004; Brennan et al. 1991). We also found that the frequency of AEs documented in studies has increased slightly over time (Figure 2); a finding that is corroborated by Danielis et al. (2021) who found an increase in the frequency of AEs from 2013-2017. Overall, the published literature suggests that one in four critically ill patients will experience an AE during their ICU stay and that the incidence may be increasing.
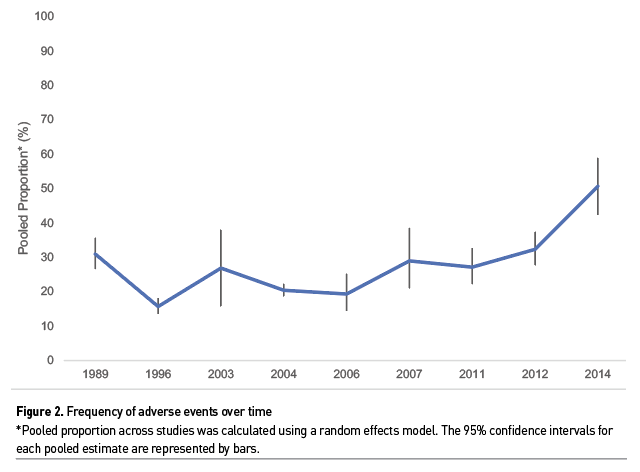
The overall frequency of AEs varied considerably between studies (range = 1.7-50.7) as did the type and frequency of each type of AE. There are several reasons for heterogeneity between study estimates, including differences in eligibility criteria, definition of AE, data collection methods as well as patient, ICU, and hospital factors. Regardless of between study variability, AEs among critically ill patients remains an important clinical problem. Exploring factors that contribute to the high rate of AEs in ICUs is needed to strategically develop evidence-based interventions to improve patient safety.
What is the Nature of AEs in the ICU?
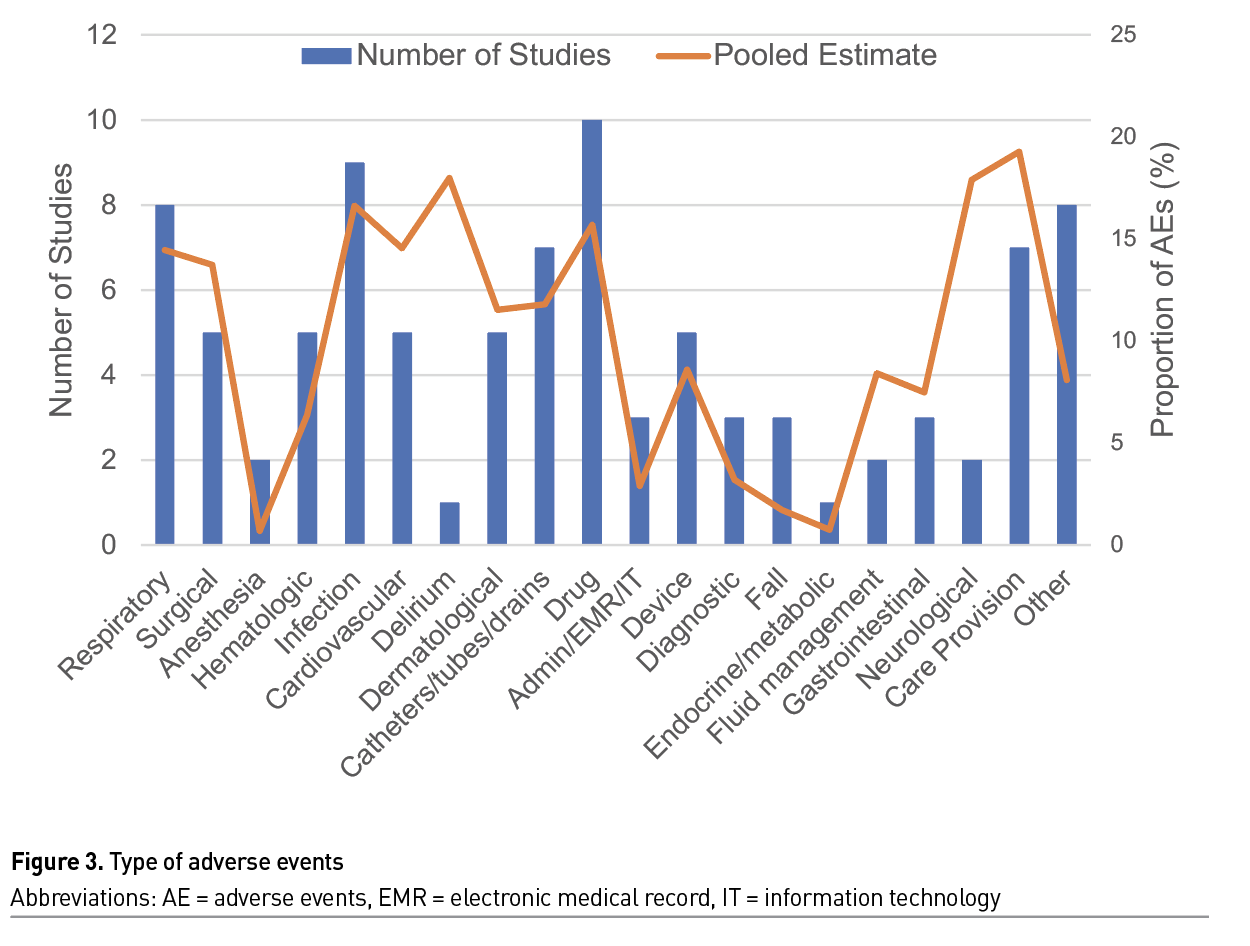
Understanding the type of AEs that occur in the ICU can help inform quality improvement initiatives. Thirteen of the 16 studies reported the type of AEs examined; 10 studies examined drug-related AEs, nine examined nosocomial infections, and eight examined respiratory AEs (Figure 3). There was considerable variation in how AEs were categorised in the absence of an accepted taxonomy of types of AEs. The incidence of each type of AE varied considerably, with the most common types of AE described related to failures in care provision which includes procedure and care management AEs (pooled estimate = 19.27% of AEs), delirium (pooled estimate = 17.97% of AEs), and neurological AEs (17.27% of AEs).
Can We Predict Which ICU Patients Will Experience an AE?
Evidence suggests several patient-level factors increase the risk of AEs in hospitalised patients; age, multimorbidity, surgical interventions, and disease severity (Roque and Melo 2016; Sauro et al. 2020a; Serafim et al. 2017; Valentin et al. 2006; Sauro et al. 2020b). We found similar factors predict AEs among critically ill patients in the ICU (Table 1). A prospective observational study of ICUs from 29 countries, identified disease severity (sequential organ failure assessment [SOFA] score and organ failure) and complexity of care (mechanical ventilation, dialysis and intravenous medication) predicted the occurrence of AEs (Valentin et al. 2006). Similarly, other studies found that older patients with more comorbidities who were either admitted to the ICU from the operating room (surgical patients) or urgently admitted to the ICU were more likely to experience an AE (Sauro et al. 2020a; Serafim et al. 2017). These studies suggest that patient-level factors can identify ICU patients at risk of experiencing AEs. However, Pronovost et al. (2006) have reported that within ICUs patient-level factors contribute to 32% of AEs, while environmental factors contribute to 22% of AEs. ICU-level factors associated with AEs include duration of ICU stay and patient to nurse ratio (Valentin et al. 2006). While these factors have been found to be associated with the occurrence of AEs, can we move beyond association to causation?
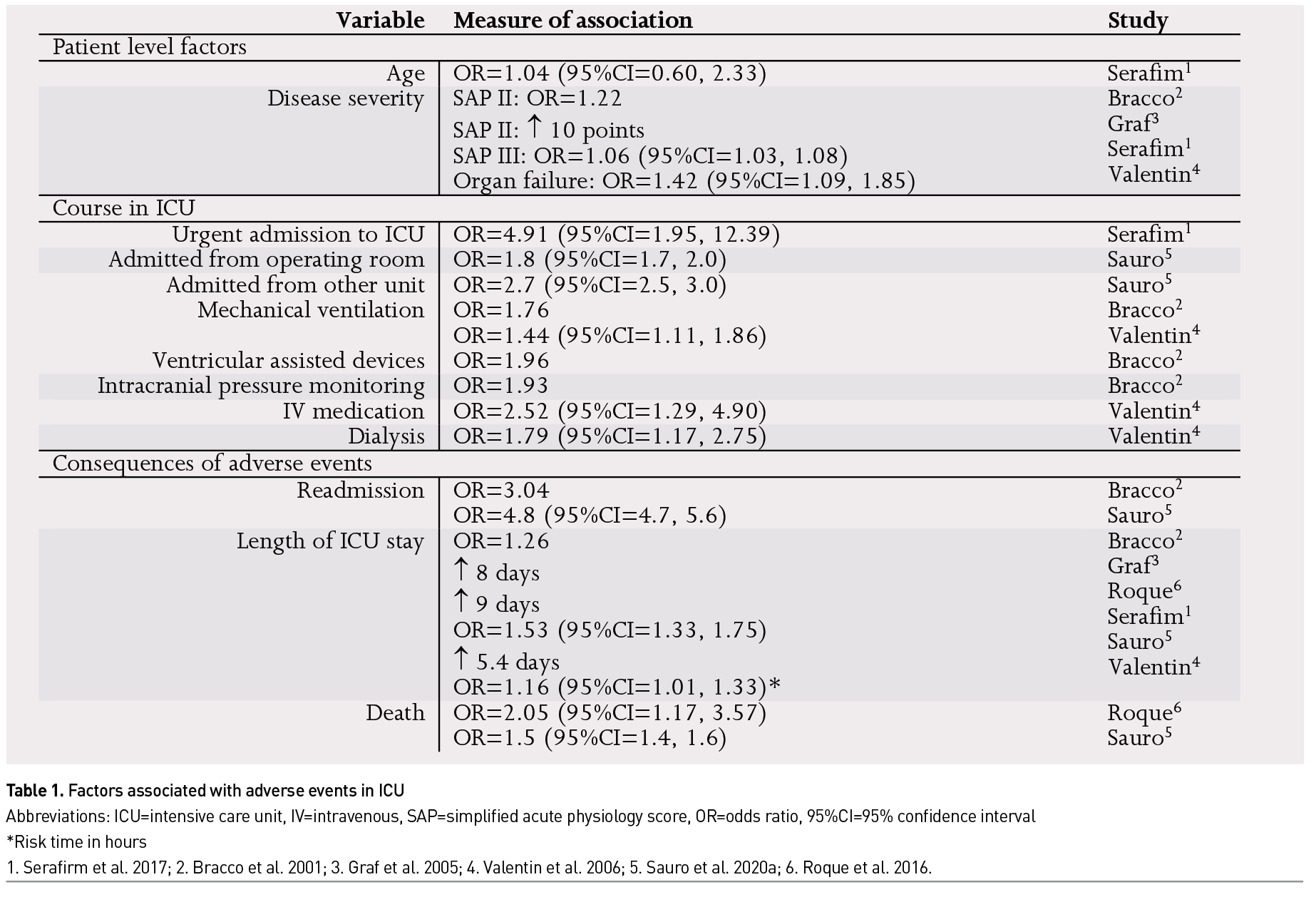
Identifying the root cause of AEs is labour intensive and challenging; consequently, there are few published studies. Bracco et al. (2001) examined human factors as the root cause of AEs in ICUs and found that human error was responsible for 31% of AEs, most commonly due to planning (wrong plan to achieve clinical goals), execution (failure to execute clinical plan as intended) and surveillance (failure to identify a change in the clinical status) failures. Of the human error-related AEs, 26% prolonged the ICU length of stay and increased the duration of patients’ stay by 15% (Bracco et al. 2001). Other factors that have been found to be associated with the occurrence of AEs in the ICU include training and education of healthcare providers (e.g., knowledge, skills, competency), team factors (e.g., communication between care providers), institutional environment (e.g., physical and human resources, workload), and information technology/electronic medical records (e.g., availability and usability of electronic resources) (Pronovost et al. 2006).
What are the Consequences of AEs in Critically Ill Patients?
Adverse events have a profound impact on patients and healthcare systems. Studies have found that AEs are associated with mortality, increased length of hospital and ICU stay and cost (Bracco et al. 2001; Graf et al. 2005; Kaushal et al. 2007). Rothschild et al. (2005), in a prospective observational study of two 10 bed ICUs (one medical and one cardiovascular), found that 12% of AEs were life-threatening and 2% resulted in death. Similar estimates were reported by Giraud et al. (1993) and Thomas et al. (2012). Garrouste et al. (2008) found that 4% of patients that experienced an AE had a prolonged ICU stay, and nearly 10% resulted in minor morbidity. Roque et al. (2016) found that patients who experienced an AE were twice as likely to die in hospital than those who did not experience an AE. Similar estimates by other authors have highlighted the increased risk of death associated with AEs (Ahmed et al. 2015; Forster et al. 2008; Roque et al. 2016; Sauro et al. 2020a).
In addition to the human cost, AEs are also costly to healthcare systems. Adverse events in the ICU increase the length of a patient’s ICU stay with estimates ranging from on average an additional 2.4 days to 31 days (Ahmed et al. 2015; Forster et al. 2008; Roque et al. 2016). In a sub-analysis of the Critical Care Safety Study conducted between 2002 and 2003, AEs were estimated to result in additional costs of $3961 USD per patient. This translated into an additional $853,000 USD per year for a ten bed medical ICU and $630,000 USD for a ten bed cardiac ICU (Kaushal et al. 2007).
These data demonstrate that the impact of AEs in critically ill patients is substantial, justifying a reinvestment in efforts to reduce AEs and improve patient safety in ICUs.
Can We Reduce AEs and Improve the Safety of ICUs?
Since the Institute of Medicine’s call to action to improve patient safety in 2000 and the establishment of the World Alliance for Patient Safety by the World Health Organization in 2004, there has been an increase in the number of studies examining AEs (Sauro et al. 2020c). We found a similar trend in the critical care literature with all but two of the 16 studies included in this review published after 2004. More worryingly, the growing number of studies does not appear to translate into fewer AEs. We might be getting better at reporting AEs, but patient safety still needs to improve.
Many AEs are considered preventable; the two studies that reported estimates of preventability found, on average, 43% of AEs were preventable (Forster et al. 2008; Rothschild et al. 2005). This would suggest a large opportunity to reduce AEs. How do we do this? Based on the available data there are two complimentary approaches to reducing AEs: (1) improving patient safety culture and its constituent organisational components and (2) targeting specific high-risk circumstances.
Several studies beyond those included in this review have examined strategies to improve patient safety in ICUs. For example, the Harvard Work Hours and Health Study found that reducing interns’ ICU shift hours reduced serious AEs by 22% (Landrigan et al. 2004). Targeted initiatives have also been successful. For example, a multicentre study in the United States implemented a multifaceted strategy to reduce the specific AE of bloodstream infections. The intervention resulted in a decrease of bloodstream infections from 7.7% to 0%, and the results were sustained for 18 months (Pronovost et al. 2010). In both examples the interventions modified structural or organisational factors related to patient safety. There is emerging evidence that suggests system-level factors, such as patient safety culture, play a significant role in the frequency of AEs (Mardon et al. 2010; Kline et al. 2008; Wang et al. 2014). Pronovost et al. (2006) reported that many factors that contributed to AEs in the ICU are structural including institutional environment. The novel COVID-19 pandemic has stressed ICU capacity in many countries and jurisdictions. Evidence suggests that physician burnout and ICU capacity strain are risk factors for preventable AEs that warrant further exploration (Sauro et al. 2020b; Panagioti et al. 2018).
Targeted approaches to reduce AEs could focus on factors associated with the occurrence of AEs or specific types of AEs. Forster et al. (2008) explored which type of AEs in ICUs are preventable and found that procedural AEs were most likely to be preventable (35% were preventable) followed by therapeutic errors (22% were preventable), while surgical complications were least likely to be preventable. There may be value to selectively targeting improvement interventions to these and other AEs that are preventable.
Given the available evidence, it is clear that patient safety (as measured by AEs) continues to be a profound challenge for ICUs and requires urgent attention. We cannot be content with the current state of AEs in ICU. It is time for us to build on the work launched by the Institute of Medicine Report To Err is Human over twenty years ago and redouble our efforts to improve patient safety.
Resolution of Case
Our case highlights the risks of AEs and their potentially serious consequences in critical care. First, the patient had a nasogastric tube inserted; procedures are associated with an increased risk of preventable AEs (Forster et al. 2008). Second, transition of the patient’s care from the ICU to a medical ward provided an opportunity for continuity of care to break down (Sauro et al. 2020b). Third, the ICU was experiencing capacity strain at the time of the transition of care, a factor associated with an increased risk of AEs (Sauro et al. 2020b), so that while a chest x-ray had been performed demonstrating the nasogastric tube to be located in the left main stem bronchus, the medical team did not review the images or radiology report. Finally, the patient was cared for by an operating room nurse redeployed to the medical unit due to pandemic staffing shortages who was unfamiliar with the standard operating procedures of the unit and restarted the patient’s tube feeds. The error was disclosed to the patient’s family. The patient received 48 hours of invasive mechanical ventilation. There was no evidence of hospital acquired pneumonia, and the patient was moved back to the medical unit after five days of further care that included chest physiotherapy.
In this case, potential prevention strategies could have included the following. First, educating healthcare practitioners and managers about the risk of AEs during periods of capacity strain when individuals may be stressed and distracted might have delayed the semi-elective reinsertion of the nasogastric tube (Bagshaw et al. 2017; Bagshaw et al. 2018). Second, implementing a forced function protocol for radiology consultants to immediately contact the most responsible health care provider for critical diagnostic findings could have resulted in earlier identification and removal of the incorrectly placed nasogastric tube by the clinical team. Third, a standardised multimodal transition in care communication (i.e., verbal and written) procedure that ensures important information including the locations and uses of tubes and lines is transmitted to the receiving care team may have prevented the reinstitution of patient’s feeds prior to confirmation of the nasogastric tube’s location (Stelfox et al. 2017).
Conflict of Interest
The authors have no conflicts of interest to report.
References:
Ahmed AH, Giri J, Kashyap R et al. (2015) Outcome of adverse events and medical errors in the intensive care unit: a systematic review and meta-analysis. Am J Med Qual. 30(1):23-30.
Bagshaw SM, Wang X, Zygun DA et al. (2018) Association between strained capacity and mortality among patients admitted to intensive care: A path-analysis modeling strategy. J Crit Care. 43:81-7.
Bagshaw SM, Opgenorth D, Potestio M et al. (2017) Healthcare Provider Perceptions of Causes and Consequences of ICU Capacity Strain in a Large Publicly Funded Integrated Health Region: A Qualitative Study. Crit Care Med. 45(4):e347-e56.
Baker GR, Norton PG, Flintoft V et al. (2004) The Canadian Adverse Events Study: The incidence of adverse events among hospital patients in Canada. CMAJ. 170(11):1678-86.
Bracco D, Favre JB, Bissonnette B et al. (2001) Human errors in a multidisciplinary intensive care unit: a 1-year prospective study. Intensive Care Med. 27(1):137-45.
Brennan TA, Leape LL, Laird NM et al. (1991) Incidence of adverse event and negligence in hospitalized patients. Results of the Harvard Medical Practice Study I. New England Journal of Medicine. 324(6):370-6.
Danielis M, Bellomo F, Farneti F, Palese A (2021) Critical incidents rates and types in Italian Intensive Care Units: A five-year analysis. Intensive Crit Care Nurs. 62:102950.
Forster AJ, Kyeremanteng K, Hooper J et al. (2008) The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res. 8:259.
Garrouste Orgeas MT, Timsit JF, Soufir L et al. (2008) Impact of adverse events on outcomes in intensive care unit patients. Critical Care Medicine. 36(7):2041-7.
Garry DA, McKechnie SR, Culliford DJ et al. (2014) A prospective multicentre observational study of adverse iatrogenic events and substandard care preceding intensive care unit admission (PREVENT). Anaesthesia. 69(2):137-42.
Giraud T, Dhainaut JF, Vaxelaire JF et al. (1993) Iatrogenic complications in adult intensive care units: a prospective two-center study. Crit Care Med. 21(1):40-51.
Graf J, von den Driesch A, Koch KC, Janssens U (2005) Identification and characterization of errors and incidents in a medical intensive care unit. Acta Anaesthesiol Scand. 49(7):930-9.
Institute of Medicine. (2000) To Err Is Human: Building a Safer Health System. Kohn LT, Corrigan JM, Donaldson MS, editors. Washington, DC: The National Academies Press. p. 312
Kaushal R, Bates DW, Franz C et al. (2007) Costs of adverse events in intensive care units. Crit Care Med. 35(11):2479-83.
Kline TJ, Willness C, Ghali WA (2008) Determinants of adverse events in hospitals--the potential role of patient safety culture. J Healthc Qual. 30(1):11-7.
Landrigan CP, Rothschild JM, Cronin JW et al. (2004) Effect of reducing interns' work hours on serious medical errors in intensive care units. N Engl J Med. 351(18):1838-48.
Mardon RE, Khanna K, Sorra J et al. (2010) Exploring relationships between hospital patient safety culture and adverse events. J Patient Saf. 6(4):226-32.
Merino P, Álvarez J, Cruz Martín M et al. (2012) Adverse events in Spanish intensive care units: the SYREC study. Int J Qual Health Care. 24(2):105-13.
Panagioti M, Geraghty K, Johnson J et al. (2018) Association Between Physician Burnout and Patient Safety, Professionalism, and Patient Satisfaction: A Systematic Review and Meta-analysis. JAMA Internal Medicine. 178(10):1317-31.
Pronovost PJ, Goeschel CA, Colantuoni E et al. (2010) Sustaining reductions in catheter related bloodstream infections in Michigan intensive care units: observational study. BMJ (Clinical research ed). 340:c309.
Pronovost PJ, Thompson DA, Holzmueller CG et al. (2006) Toward learning from patient safety reporting systems. Journal of Critical Care. 21(4):305-15.
Resar RK, Rozikh JD, Simmonds T et al. (2006) A trigger tool to identify adverse events in the intensive care unit. Joint Commission journal on quality and patient safety/Joint Commission Resources. 32(10):585-90.
Roque KE, Tonini T, Melo EC (2016) Adverse events in the intensive care unit: impact on mortality and length of stay in a prospective study. Cadernos de saude publica. 32(10):e00081815.
Rothschild JM, Landrigan CP, Cronin JW et al. (2005) The Critical Care Safety Study: The incidence and nature of adverse events and serious medical errors in intensive care. Crit Care Med. 33(8):1694-700.
Sauro KM, Machan M, Whalen-Browne L et al. (2021) Evolving Factors in Hospital Safety: A Systematic Review and Meta-Analysis of Hospital Adverse Events. J Patient Saf. 17(8):e1285-e95.
Sauro KM, Soo A, Quan H, Stelfox HT (2020a) Adverse Events Among Hospitalized Critically Ill Patients: A Retrospective Cohort Study. Med Care. 58(1):38-44. (18)
Sauro KM, Soo A, de Grood C, Yang MMH et al. (2020b) Adverse Events After Transition From ICU to Hospital Ward: A Multicenter Cohort Study. Crit Care Med. 48(7):946-53. (24)
Sauro K, Ghali WA, Stelfox HT et al. (2020b) Measuring safety of healthcare: an exercise in futility? BMJ Qual Sal. 29(4):341-4. (26)
Sauro KM, Quan H, Sikdar KC et al. (2017) Hospital safety among neurologic patients: A population-based cohort study of adverse events. Neurology. 89(3):284-90.
Serafim CTR, Dell'Acqua MCQ, Castro M et al. (2017) Severity and workload related to adverse events in the ICU. Rev Bras Enferm. 70(5):942-8.
Stelfox HT, Leigh JP, Dodek PM et al. (2017) A multi-center prospective cohort study of patient transfers from the intensive care unit to the hospital ward. Intensive Care Med. 43(10):1485-94.
Thomas AN, Taylor RJ (2012) Review of patient safety incidents reported from critical care units in North-West England in 2009 and 2010. Anaesthesia. 67(7):706-13.
Valentin A, Capuzzo M, Guidet B et al. (2006) Patient safety in intensive care: Results from the multinational Sentinel Events Evaluation (SEE) study. Intensive Care Medicine. 32(10):1591-8.
Zegers M, De Bruijne MC, Spreeuwenberg P et al. (2011) Variation in the rates of adverse events between hospitals and hospital departments. Int J Qual Health Care. 23(2):126-33.
Wang X, Liu K, You LM et al. (2014) The relationship between patient safety culture and adverse events: a questionnaire survey. Int J Nurs Stud. 51(8):1114-22.





















