ICU Management & Practice, ICU Volume 13 - Issue 1 - Spring 2013
A Compartment Syndrome (CS) is Defined as Increased Pressure in a Closedanatomic space which threatens the viability
of enclosed and surrounding tissue (Malbrain et al. 2006). Within the body
there are four major compartments: the head, the chest, the abdomen and the
extremities. Within each compartment, individual organs can be affected by a
CS. As such, orbital CS, cardiac CS, hepatic CS, renal CS and pelvic CS have
all been described separately within this paper. This review will discuss the
different aspects of PCS and the interactions between individual compartments.
Introduction
A CS
can have many causes and can develop within many disease processes, but most
importantly it is often related to massive fluid loading in the setting of
capillary leak leading to second and third spacing of fluids. This fluid
accumulation will lead to organ oedema and organ dysfunction. The abdominal
compartment has unique anatomical properties because it is “up-stream” from the
extremities and “downstream” from the chest. Therefore, it may influence the
pathophysiology of other compartments as well. The presence of a CS often plays
a role when we are dealing with therapeutic dilemmas or conflicts. A
therapeutic conflict is a situation where each of the possible therapeutic
decisions carries some potential harm. In high-risk patients, the decision
about fluid administration is taken within this context. Therapeutic conflicts
are the biggest challenge for protocolised cardiovascular management in
anaesthetised and critically ill patients, where our decisions can make the
most difference (Kavanagh and Meyer 2005; Malbrain et al. 2012).
Scalea, with colleagues, was the first to introduce the term multiple
CS in a study of 102 patients with increased intra-abdominal (IAP),
intrathoracic (ITP), and intracranial pressure (ICP) after severe brain injury
(Scalea et al. 2007). He suggested that different compartments within the body
are not isolated and independent entities but instead are closely connected.
Because the term multiple CS is nowadays mostly used in relation to multiple
limb trauma with CS, needing fasciotomy, the term polycompartment syndrome
(PCS) was finally coined in 2007 and proposed in order to avoid confusion
(Malbrain and Wilmer 2007). Organ-organ interactions, and thus also the
presence of a PCS, may occur more often than we realise in real life.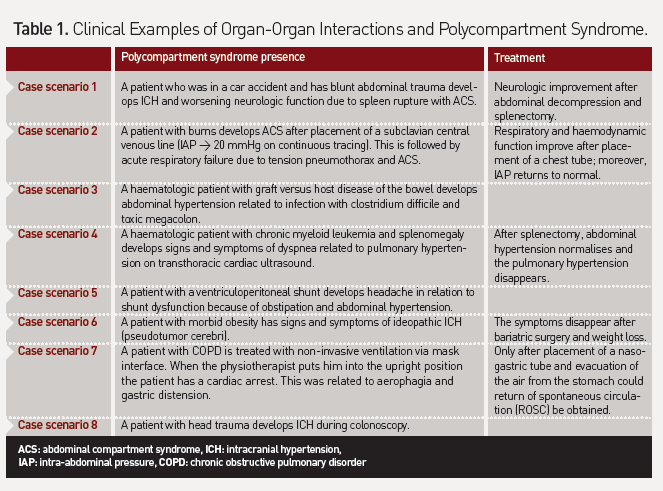
Pathophysiology of the Four Major Compartment Syndromes
An increased compartment pressure (CP) will increase venous resistance and decrease perfusion pressure in the implicated compartment, but will also affect other compartments. The resulting impact on end-organ
function
and viability can be devastating.
1. Intracranial Compartment Syndrome
Any change in volume in the head leads to a reciprocal change in
the size of the remaining components, and results in an increase in ICP and a
decrease in cerebral perfusion pressure (CPP), defined as mean arterial
pressure (MAP) minus ICP (CPP=MAP–ICP). Treatment options for intracranial hypertension
(ICH) either focus on lowering ICP or raising CPP. However, fluid therapy used
to support CPP may exacerbate visceral oedema, promote ascites and increase
IAP, which in turn can further increase ICP (De Laet et al. 2007). In patients
with severe traumatic brain injury, treatment decisions may result in a vicious cycle that increases pressures in various
compartments (Scalea et al. 2007). Therefore, PCS should be considered present
in patients with multiple injuries, with increased ICP, who do not respond to
conventional therapy (Scalea et al. 2007).
2. Thoracic Compartment Syndrome
Thoracic CS (TCS) has traditionally been described in patients undergoing cardiac surgery (Kaplan et al. 1996; Rizzo and Sample 2003). TCS is rare in patients with thoracic trauma due to the limited survival when injuries are significant enough to result in massive tissue oedema after resuscitation. In the ICU, increased intrathoracic pressure (ITP) is most commonly related to sepsis, capillary leak, fluid resuscitation, positive pressure ventilation with high positive end-expiratory pressure (PEEP) or dynamic hyperinflation, pneumothorax, COPD with auto-PEEP, diminished chest wall compliance (e.g. morbid obesity or eschars), lung fibrosis and ARDS. Rising ITP and mean or peak inspiratory pressures during thoracic wall closure may serve as an early warning that a patient is at risk for TCS. Since increased ITP, like raised IAP, is most commonly related to excessive fluid resuscitation both frequently coexist (Talmor et al. 2006; Valenza et al. 2007). Some key issues to remember are: best PEEP should be set to counteract ITP and IAP whilst at the same time avoiding over-inflation of already well-aerated lung regions (best PEEP (cmH2O)=IAP (mmHg)). During lung-protective ventilation, plateau pressures (Pplat)
should
be limited to transmural plateau pressures (Pplattm) below 35 cmH2O (Pplattm=Pplat–ITP=Pplat IAP/2<35cmH2O). Increased ITP and IAP facilitate lung
oedema. Therefore, monitoring of extravascular lung water index (EVLWi) could
be beneficial (Quintel et al. 2004). Spontaneous respiration and mechanical
ventilation increase inspiratory and decrease expiratory IAP. Therefore, ΔIAP may indirectly predict abdominal wall
compliance (Figure 2) (Malbrain and Wilmer 2007; Sturini et al. 2008).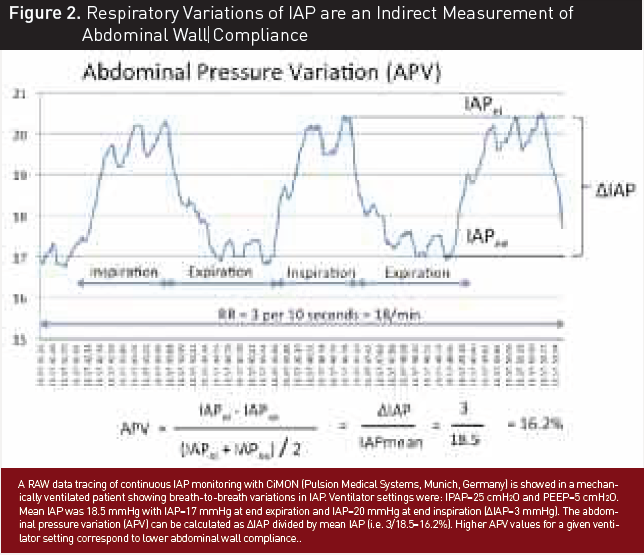
3. Abdominal Compartment Syndrome
The abdomen can be considered as a closed box with partially rigid
sides (spine, pelvis and costal arch) and partially flexible sides (abdominal
wall and diaphragm). Since the abdominal cavity can be considered as a
relatively non-compressible and primarily fluid containing compartment,
behaving in accordance to Pascal’s law, the IAP measured at one point can be
assumed to represent the IAP throughout the entire abdomen (Malbrain 2004;
Malbrain and Jones 2006). In normal conditions, IAP ranges from 0–5 mmHg
(Sanchez et al. 2001; De Keulenaer et al. 2009). The gold standard IAP
measurement method is via the bladder, either by a transurethral Foley Manometer
(Holtech Medical), or coupled to an AbViser valve (ConvaTec), or any homemade
system (Malbrain 2004). The term abdominal CS (ACS) was first used by Fietsam
and colleagues in the late 1980s to describe the pathophysiologic alterations
resulting from IAH secondary to aortic aneurysm surgery (Fietsam
et al. 1989). The World Society on Abdominal Compartment Syndrome (WSACS –
www.wsacs.org) recently published consensus
definitions (Malbrain et al. 2007). Analogous to the widely accepted and clinically
utilised concept of CPP, abdominal perfusion pressure (APP) has been proposed as
a more accurate predictor of visceral perfusion and a
potential endpoint for resuscitation by considering both arterial inflow (MAP)
and restrictions to venous outflow (IAP) (APP=MAP–IAP) (Cheatham et al. 2000;
Malbrain 2002; Deeren et al. 2005; Cheatham and Malbrain 2006).
4. Limb or Extremity Compartment Syndrome
Extremity CS (ECS) is a condition in which the pressure within a closed muscle compartment increases to a level that reduces capillary blood perfusion below the level necessary for tissue viability. Permanent loss of function and contracture may occur. Extremity CP can be measured by a needle connected to a fluid-filled pressure transducer system. Normal CP values should be <20 mmHg. This technique can be used to guide the need for surgical intervention: tissue perfusion pressure should be equal to capillary pressure minus extremity CP. ECS is especially common in obese patients and mostly results from trauma with fractures (especially of the tibia), tight plaster casts, muscle contusions, bleeding disorders, burns (with eschars), venous obstruction and arterial occlusion with post-ischaemic swelling. ECS will result in muscle compression and rhabdomyolysis, which may cause hypovolemia, acute kidney injury (AKI), coagulopathy, acute lung injury (ALI) and shock. Therefore, ECS may also have distant effects on other organs (Figure 1). In case of established ECS, the only definitive treatment is decompressive fasciotomy with muscle debridement in case of necrosis. Apart from its influence on other distant organs, extremity CP is influenced itself by increased IAP related to ACS or Pelvic CS, as both conditions diminish venous return from the extremities, promoting further limb swelling.
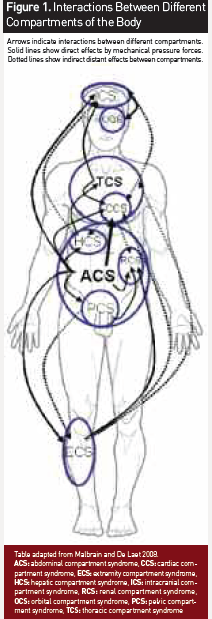
Pathophysiology of Subsidiary Compartment Syndromes
1. Orbital Compartment Syndrome
Acute orbital CS (OCS) is a rare complication of increased pressure within the confined orbital space. An increased intraorbital pressure (IOP) may cause decreased orbital perfusion pressure (OPP) by a mechanism similar to mass lesions (OPP=MAP–IOP). OCS presents with typical signs and symptoms (eye pain, reduced ocular motility, pro-optosis, diplopia) and progressive visual deficits. Recognition and prompt treatment is of paramount importance to prevent blindness. A recent study in burn patients showed that increased IOP was significantly associated with the amount of fluids given during the first 24 hours of hospitalisation and with the presence of peri-ocular burns (Singh et al. 2008).
2. Cardiac Compartment Syndrome
Within the thorax, cardiac tamponade can be considered as a specific CS. Strikingly, as little as 250 ml of fluid can cause acute cardiac tamponade. However, under chronic conditions, much greater amounts of fluid can accumulate as the cardiovascular system can slowly adjust. A similar condition arises when either ITP directly (in case of TCS), or IAP indirectly (in case of ACS) compresses the cardiac chambers. The latter is due to an upward movement of the diaphragm. In case of increased ITP or IAP, coronary perfusion pressure (CoPP) is lowered (CoPP=DBP–PAOP=DBP–ITP) (where DBP is diastolic blood pressure and PAOP is pulmonary artery occlusion pressure). Increased ITP also results in a more difficult preload assessment because invasively measured filling pressures will be falsely increased. Transmural filling pressures, calculated as the end-expiration value minus ITP may better reflect preload (Valenza et al. 2007; Ridings et al. 1995) (CVPtm=CVPee–ITP or PAOPtm=PAOPee–ITP). A quick estimate of transmural filling pressures can also be obtained by subtracting half of the IAP from the end-expiratory filling pressure (Wauters et al. 2007) (CVPtm= CVPee–IAP/2 or PAOPtm=PAOPee– IAP/2). “Volumetric” preload estimates such as right ventricular end diastolic volume index (RVEDVi) or global end diastolic volume index (GEDVi) are useful alternatives for pressure- based measurements in conditions of ITP (Malbrain and Cheatham 2004; Schachtrupp et al. 2003; Michard et al. 2003).
3. Hepatic Compartment Syndrome
Because the liver is an encapsulated organ, local haematoma formation caused by trauma or bleeding diathesis (oral anticoagulants, liver cirrhosis, and so on) may compromise tissue perfusion by causing hepatic CS (HCS) (Goldman et al. 2003; Pearl and Trunkey 1999).
4. Renal Compartment Syndrome
The association between IAH and renal impairment has been known for over 150 years (Schein 2006). However, the exact (patho)physiological interplay between IAP and kidney injury has only been studied intensively in recent years (Biancofiore et al. 2003; Sugrue et al. 2006; De Laet et al. 2007). Elevated IAP significantly decreases renal blood flow and causes renal venous hypertension through pressure transduction, leading to renal dysfunction and failure (Kirkpatrick et al. 2006; Wauters et al. 2009). Oliguria develops at IAP>15 mmHg and while values >25 mmHg are associated with anuria in the presence of normovolemia. Within the capsule of the kidney itself, local haematoma formation (caused by trauma or bleeding diathesis) may have a further adverse effect on tissue perfusion, causing a local renal compartment syndrome.
5. Pelvic Compartment Syndrome
In the pelvic region, three major compartments— gluteus medius-minimus, gluteus maximus, and iliopsoas—can be distinguished from the smaller compartment of the tensor fasciae latae muscle. Pelvic CS (PCS) is rare and a clear history of trauma is often lacking (Bosch and Tscherne 1992; Hessmann and Rommens 1998). It is often associated with drug and alcohol abuse, infections (necrotising fasciitis) and the use of anticoagulant therapy (Hessmann and Rommens 1998). Increased PCP may eventually increase IAP and affect kidney function due to bilateral ureteral obstruction. Moreover, massive intrapelvic haematoma with increased retroperitoneal pressure can cause renal failure. Decompressive fasciotomy of the gluteal compartment is the treatment of choice.
6. Cardio-Abdomino-Renal Syndrome
The abdominal compartment could potentially form a missing link in the pathophysiology of acute decompensated heart failure (ADHF) and cardiorenal syndrome. Only recently, it was shown that raised IAP is prevalent in cases of advanced heart failure with reduced ejection fraction, and that it correlates with impairment of renal function (Mullens et al. 2008). Importantly, medical treatment resulting in a decrease of IAP ameliorates renal function, and in cases of persistent high IAP, ultrafiltration might be beneficial (Mullens et al. 2008). Notably, while organ dysfunction in intensive care literature has only been described when IAP exceeds 12 mmHg, patients with ADHF already develop worsening renal function with a much lower IAP (Mullens et al. 2008). This might suggest that the underlying reserve of the kidneys to counteract increased IAP is limited in this setting. Therefore, we would like to define Cardio- Abdominal-Renal Syndrome (CARS), to emphasise the potentially important role of the abdominal compartment and splanchnic vasculature in the pathophysiology of ADHF and cardiorenal syndrome.
Clinical Management
The management of patients with PCS is based on three principles (Mayberry 2006; Parr and Olvera 2006):
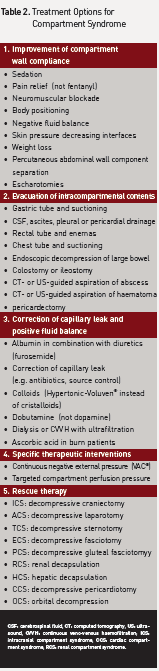
1. Specific medical and surgical procedures to reduce CP (Table 2), including:
• Improvement of compartment wall compliance;
• Evacuation of intracompartmental contents;
• Correction of capillary leak and positive fluid balance;
• Specific treatments; and
• Rescue treatments.
2. General and organ support (intensive care) of the critically ill patient.
3. Optimisation and prevention of specific adverse events after surgical decompression (ischaemia/reperfusion).
Take Home Message
While PCS is uncommon, its consequences can be significant. The abdomen plays a central role, and the effect of IAH on different organ systems has been described, along with recommendations to compensate for these effects. The abdominal compartment could play a key role in the pathophysiology of acute decompensated heart failure and cardiorenal syndrome. The ultimate treatment goal of PCS is not only to decrease CP, but also to improve organ function and to decrease mortality.



















