ICU Management & Practice, ICU Volume 14 - Issue 2 - Summer 2014
Early Recognition of Sepsis
Patients with sepsis, now defined as a severe infection with some degree of associated organ dysfunction, make up a large proportion of the critically ill population and, although outcomes have improved over the last decade, mortality rates remain above 20-30%, and even close to 40% when shock is present. There are no effective specific anti-sepsis treatments, and management of patients with sepsis thus relies largely on early recognition allowing rapid institution of correct therapeutic measures, including administration of appropriate antibiotics, source control measures when necessary and effective resuscitation strategies with intravenous fluids and vasoactive agents when required. Additional therapies, such as corticosteroids or other immunomodulatory drugs, have a limited place (especially after the withdrawal of activated protein C from the drug market).
Early and aggressive management with appropriate antimicrobials and rapid and complete haemodynamic stabilisation has been shown to be associated with improved outcomes. The ability to accurately diagnose sepsis as soon as possible is, therefore, paramount. However, it is difficult to know in advance who will develop sepsis. A minor infection (even an apparently simple viral cold) can evolve into sepsis, but (fortunately!), not all people with minor infections will develop organ dysfunction and sepsis. Although advances in genomics and proteomics are providing some clues as to which patients may be more likely to develop sepsis than others, these techniques remain exploratory at present. For many years some people used the so-called systemic inflammatory response syndrome (SIRS) criteria (hypo- or hyperthermia, tachycardia, tachypnea, altered white blood cell count) to screen patients for possible sepsis, but these criteria are present in most critically ill patients, so have limited use in trying to distinguish those who have or will develop sepsis from other patients. These criteria are currently used to reflect the presence of infection and this is important; however, fever and other signs of infection are commonly present in acutely ill patients, and these patients are frequently already receiving antibiotics, such that microbiological cultures will be negative in as many as 50%. Imaging modalities to locate a source are also often unreliable, e.g. lung opacities may be due to infection, but could also be the result of atelectasis, or lung oedema due to acute respiratory distress syndrome (ARDS) or acute heart failure. Improved bioinformatics and microarray techniques may improve our ability to identify the presence of microorganisms, but these techniques are not yet available for routine clinical use. Various biomarkers have also been suggested to help in the diagnosis of sepsis, e.g. procalcitonin, C-reactive protein, interleukin-6 levels, but as yet none is specific for sepsis. Panels of multiple biomarkers may be more helpful, but further research is needed to define which biomarkers should be included in such panels.
Rather than focusing on signs or markers of infection to diagnose sepsis, it is perhaps better to focus on the presence or development of organ dysfunction, and to evaluate whether this may be due to sepsis. Any organ dysfunction without a clearly identifiable cause should always raise the possibility of sepsis, but six types may be particularly indicative (see Box 1). Importantly, because of the systemic nature of the sepsis response any organ system may be affected regardless of the source of the underlying infection. Respiratory dysfunction is particularly common, and may manifest simply by tachypnea or hypoxaemia, necessitating oxygen therapy or mechanical ventilation in more severe cases. Renal dysfunction often leads to reduced urine output and later to increased serum creatinine levels. Cardiovascular dysfunction can include tachycardia and hypotension and resultant poor tissue perfusion leading to increased blood lactate levels. Abnormalities in blood clotting are also common in patients with sepsis, and include a reduced platelet count as one of the early signs. Unexplained confusion and reduced alertness are signs of central nervous system dysfunction. Jaundice with raised bilirubin levels indicates liver dysfunction, although this occurs relatively late in the process.

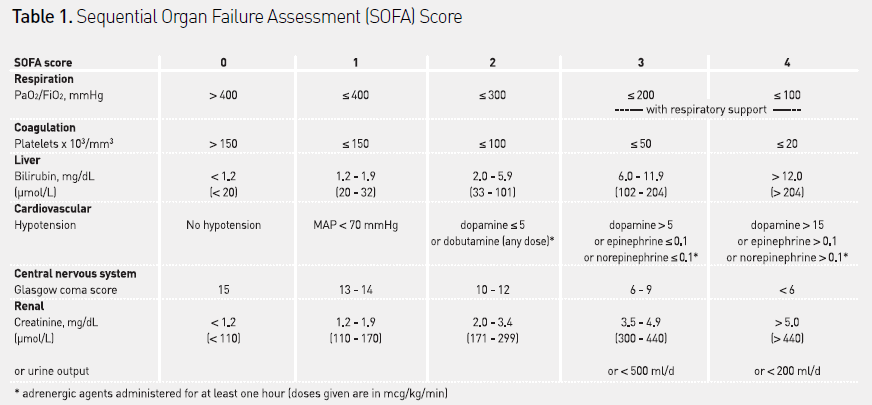
The most widely used system to assess organ dysfunction in critically ill patients is the sequential organ failure assessment (SOFA) score (see Table 1). This score quantifies organ dysfunction in six organ systems - respiratory, cardiovascular, hepatic, coagulation, renal and neurological – using a score from 1 (least dysfunction) to 4 (most dysfunction). Individual scores can be combined to give an overall indication of the degree of organ dysfunction in individual patients (range 0-24). The SOFA score on admission to the ICU is correlated with the development of bacteraemia. Higher scores are correlated with a higher risk of death, and changes in SOFA score over time can indicate response to therapy, with stable or increasing scores suggesting a worsening condition.
Increased awareness of sepsis and more rapid management of patients with sepsis have helped to improve outcomes in recent years, but mortality rates remain unacceptably high. While waiting for specific therapies to become available, continued emphasis must therefore be on the importance of early diagnosis allowing rapid infection control and organ support.
References:
Dellinger RP, Levy MM, Rhodes A et al. (2013) Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med, 39: 165-228.
Kaukonen KM, Bailey M, Suzuki S et al. (2014) Mortality related to severe sepsis and septic shock among critically ill patients in Australia and New Zealand, 2000-2012. JAMA, 311: 1308-16.
Levy MM, Fink MP, Marshall JC et al. (2003) 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med, 31: 1250-6.
Vincent JL, Moreno R, Takala J et al. (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med, 22: 707-10.
Vincent JL, Opal SM, Marshall JC, Tracey KJ (2012) Sepsis definitions: time for change. Lancet, 381: 774-5.
Vincent JL, Beumier M (2013) Diagnostic and prognostic markers in sepsis. Expert Rev Anti Infect Ther, 11: 265-75.



















