HealthManagement, Volume 21 - Issue 7, 2021
Key Points
- Patient engagement is the next blockbuster drug.
- Patient engagement has long-proved to improve patient safety, reduce hospital admissions and substantially lower use of other healthcare resources.
- There is undeniable cost savings and a huge return-on-investment (ROI) when industry adopts these programmes.
- Getting patient engagement right – from the inside out – and creating a transferable business value from patient engagement is key to making sure that a patient centric organisational mindset works – and pays.
If a pharma company had a medicine that could be developed without risk, was highly-efficacious and made significant improvements to the patient’s life, it would be a no-brainer to invest in bringing that asset through the pipeline as quickly as possible, right?
This is the assertion made by health journalist, Leonard Kish, in 2012 when he predicted that “patient engagement is the next blockbuster drug”. In fact, Kish was so convinced about the therapeutic benefits of patient engagement that he postulated that it should be routinely ‘prescribed’ as an adjuvant, such that not to provide patient engagement would be a serious breach of medical responsibility. Patient engagement has long-proved to improve patient safety, reduce hospital admissions and substantially lower use of other healthcare resources so reducing healthcare costs. When it comes to the question of ‘show me the money’ when considering investing in a long term patient engagement programme, there is undeniable cost savings and a huge return-on-investment (ROI) when industry adopts these programmes into their R&D system as early as possible.
The potential for patient engagement to become a therapy, a commodity and a catalyst to improve all aspects of pharma business and product development from that clarion call is coming to fruition. Getting patient engagement right – from the inside out – and creating a transferable business value from patient engagement is key to making sure that a patient centric organisational mindset works – and pays. Payment is in the form of the ‘triple win’ – approaches, products services and enduring policies that deliver benefits for patients, for pharma and for society.
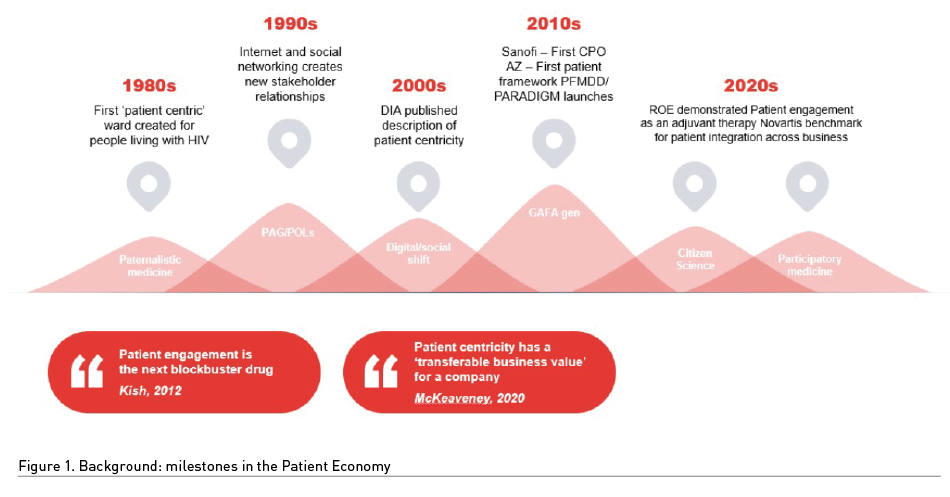
Patient Centricity Is Not An Overnight Success
Despite the intensity of patient centricity projects and the plethora of branded patient engagement programmes run by pharma, we are still struggling to justify investment in approaching patient engagement as a strategic business essential for a company. The milestones of patient engagement are given in Figure 1, behind which are inputs from broader societal change and expectations from patient and advocacy groups which started more than three decades ago with the first ‘patient centric ward 4b’ opening for people being treated in 1984 for HIV infection. There are parallels for patients living with rare conditions today to those early struggles and patient lobbying groups for people living with HIV. Both patients group share the determination to be ‘heard’ within pharma – to disrupt, contribute to and challenge clinical trial protocols – to do whatever needs to be done to get new medicines to patients as soon as possible. As such, it is evident that patient engagement impacts on every ‘department’ within pharma walls and the outcomes of patient engagement practices internally provide huge societal impact. Patient engagement therefore, must be strategically driven and recognised as requiring end-to-end practices from patient-focused drug development through to long-term patient support programmes.
Given that bottomless funding pockets do not exist and that pharma is increasingly scrutinised for the financial relationships it has with Patient Advocacy Groups, it is crucial that, internally and externally, a company has a transparent standard operating and reporting procedure for all investment in patient engagement activities. The return on investment must be demonstrated from all patient engagement projects. Again, there is nothing sinister in this reporting when it is approached from the ‘triple win’ perspective. Figure 2 summarises the current academic ‘proof’ that this triple win is achievable – and acceptable to our different healthcare regulators and society itself.
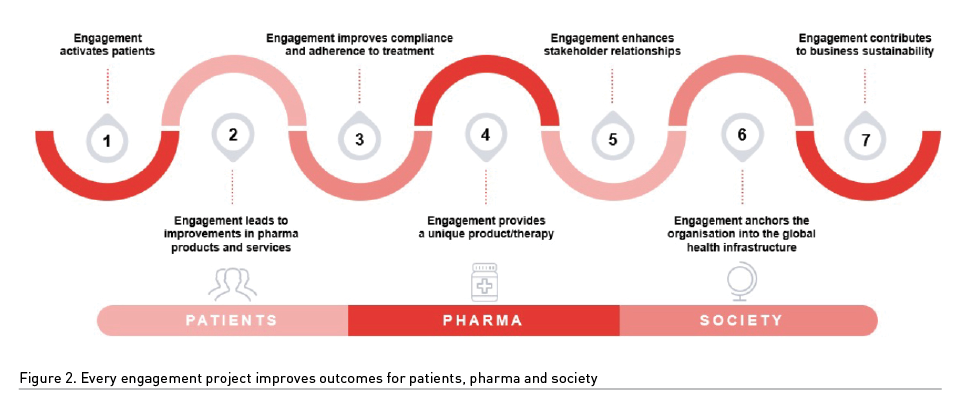
Proof That Patient Engagement Secures The ‘Triple Win’
Within a pharma company, the importance of, and needs for, patient engagement differs according to the department (Figure 3). This serves to demonstrate that an overall patient centric approach for an organisation is essential.
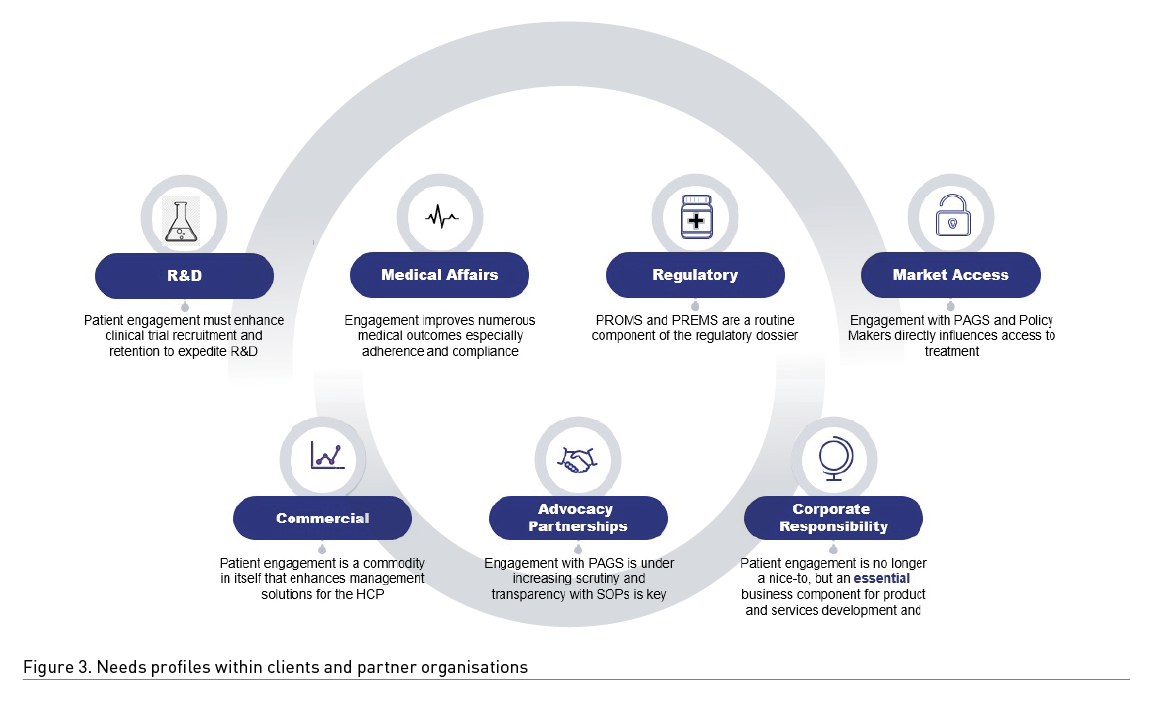
The pivotal ‘relationship vignettes’ that require a strategic approach to patient engagement are as follows:
- Product development depends on clinical trial participation – clinical trial participation depends on engaged patients being recruited and staying in the trial – the clinical trial protocol needs to be practically and psychologically percipient of the patient’s needs and expectations and this relationship must be established as early as possible to build trust and collaboration.
- Patient advocacy and lobbying for early diagnosis and to truncate the patient journey can be assisted by pharma-sponsored awareness and patient educational programmes.
- Regulatory approval routinely expects data from Patient Reported Outcome Measures and Patient Reported Experience Measures – PROMS and PREMS – are expected to be part of clinical trial protocols by expert patients. Again, including patients in the creation of PROMS and PREMS requires early and enduring relationships.
- Access to treatment and care is competently improved and expedited when pharma and patient groups collaborate to highlight unmet medical needs.
- Long term benefits of treatment – adherence and compliance and patients who are motivated in self-care contributions to their wellbeing have a measurable reduced use of other healthcare resources which is of great benefit to society.
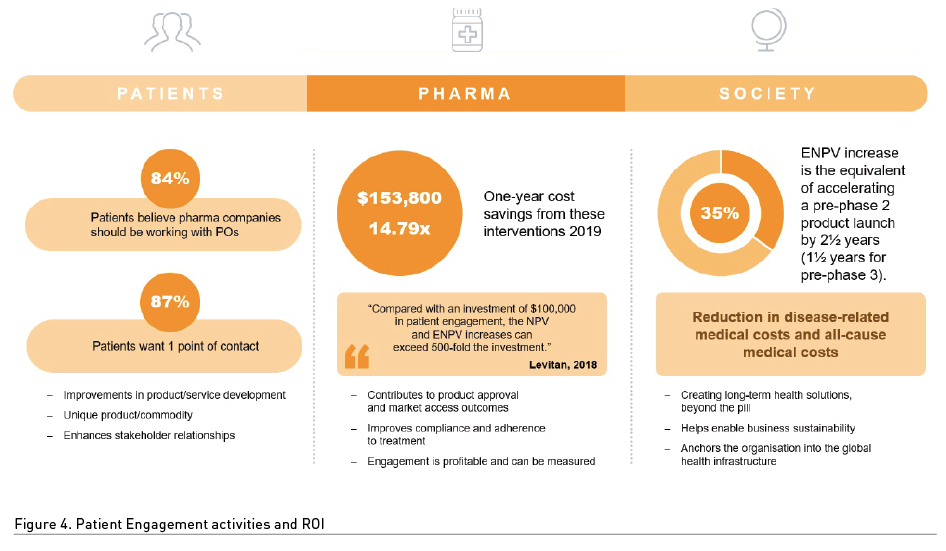
Companies who ‘join the dots’ as described above, within their organisation and communicate, upskill, and train employees to recognise their contribution within a patient centric approach to patient engagement to deliver the ‘triple win’ are assured of more efficient and justifiable return on investment in patient engagement initiatives. Companies who communicate this intent externally are able to improve measurement and validation of a strategic approach to patient engagement across the sector and to retain the current increased societal trust that is resulting from offering life-saving solutions in response to COVID-19 infection. The time is right for pharma to be recognised for the incredible contributions made to improving and elongating human health. The triple win attitude amplifies the importance of pharma’s role in the global healthcare infrastructure. Importantly, it can be evidenced that patient engagement activities demonstrate excellent ROI in every desired outcome – from R&D through to global sustainability in healthcare initiatives (Figure 4). Crucially, for pharma, patient centricity across the organisation – by investing in patient engagement activities in all areas of the business, ultimately has the potential to bring a product to market some two and a half years earlier and patient support programmes substantially reduce all disease related health costs. As a ‘blockbuster’ drug, therefore, it is little wonder that such invaluable return is establishing patient engagement initiatives as therapeutic/adjuvant products in their own right.
Show Me the Strategy Then Show Me the Money: PEP, PIPP and PVP
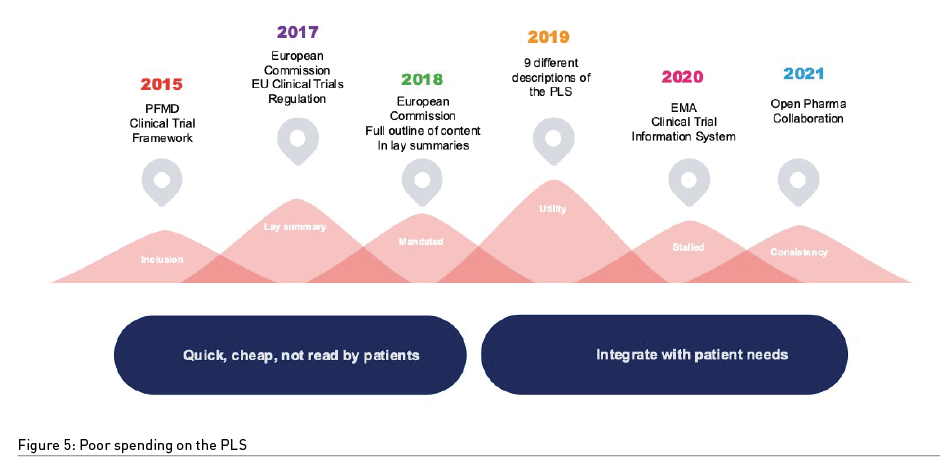
At Prime Patient we work with clients at the strategic outset to create their ‘Patient Engagement Programme’ (PEP) and to consult with all departments to identify where the ‘money saving’ aspects of patient engagement exist and where patient engagement campaigns will have the greatest ROI. This is a deep-dive, highly methodological approach which generates a ‘Patient Insights Positional Paper’ (PIPP) where our clients benchmark their patient engagement capabilities and successes to date compared with up to 10 other companies competing for the same patient-attention bandwidth. This provides clear indication of where to spend and where not to spend. As such, when an industry client approaches us with a ‘shopping list’ of patient materials we like to reassure them to take a step-back and evaluate if the individual activities will ultimately deliver the triple-win using the PIPP to facilitate decision-making. The internet is saturated with patient websites, attempts by pharma to create ‘new patient communities’ and most companies will have a pile of ‘patient journeys’ gathering dust once created. This is spend for the sake of it. Take the example of the ‘Plain Language/Patient Lay Summary’ (PLS) which has been in circulation since 2017 (Figure 5). For patients – they don’t read them, they don’t access them, they don’t share them. The PLS does little to increase patient activation or boost patient-pharma collaboration. Enter then the newly launched ‘Patient Voice Publication’ (PVP) – our answer to creating a patient-pharma connecting tool that enable patients to describe their ‘real word’ needs to pharma to improve product, service and clinical trial design, gains insights from patients for pharma and increases health literacy. This is perfect ‘triple win’ thinking … and the right spend.\
Increasingly pharma will come under scrutiny to defend their relationships with Patient Advocacy Groups and to highlight the benefits of their patient support programmes (PSPs). Elevating patient engagement to be an essential business spend for patient safety, support and societal gain is the perfect way to ‘show the benefit of the money’ invested in patient engagement programmes by pharma.
Conflict of Interest
None.
















